Abstract
Background: Domperidone is commonly used off-label to stimulate milk production in mothers who have low milk supply. The aim of this study was to describe trends, patterns and determinants of postpartum domperidone use.
Methods: This is a retrospective, population-based study involving all women with a live birth between Jan. 1, 2002, and Dec. 31, 2011, in the province of British Columbia. We examined administrative data sets containing person-specific information on filled prescriptions and use of medical services, and we used logistic regression to examine associations between domperidone use and maternal characteristics.
Results: The study population consisted of 225 532 women with 320 351 live births. The prevalence of postpartum domperidone use more than doubled between 2002 and 2011. In 2011, 1 in 3 women with a preterm birth and 1 in 5 women with a full-term birth were prescribed domperidone in the first 6 months postpartum. Women who were older, had a higher body mass index, had a chronic disease, were first-time mothers, delivered more than 1 baby (multiple pregnancy), had a preterm birth or had a cesarian delivery were more likely to fill a postpartum domperidone prescription.
Interpretation: We found an increase in postpartum domperidone use over a 10-year period. More research is needed on maternal and infant health outcomes.
Breastfeeding is strongly encouraged over formula feeding because of multiple health benefits for infants, including improved gastrointestinal function and decreased risk of acute respiratory and gastrointestinal infections as well as otitis media.1 Difficulty in establishing adequate milk supply is a common challenge, and prescription drugs may be seen as an attractive solution.
Domperidone, a dopamine antagonist available in Canada since 1985, is used to increase upper gastrointestinal motility and to prevent treatment-related gastrointestinal symptoms in patients with Parkinson disease. It also increases prolactin levels, and its use to stimulate lactation was first described more than 30 years ago.2 However, domperidone has not been approved for this indication in Canada. Yet in a recent study of prescription drug use during pregnancy and the postpartum period in British Columbia,3 we identified domperidone to be the most commonly prescribed drug during the first 6 months postpartum.
Limited evidence suggests that domperidone can increase milk supply, especially following preterm birth.4-7 A dose-response relation remains uncertain, as 2 small studies failed to indicate a clear advantage with higher doses,8,9 and large-scale studies are lacking. A multicentre trial in preterm births, still ongoing in 2015, aims to randomly assign 560 women to 2 groups and follow them for 28 days at 30 mg/d, the first 2 weeks placebo-controlled.10
Importantly, maternal and infant safety have not been established. During the 2000s, approved product information in Canada discouraged domperidone use during breastfeeding and recommended doses of 30-40 mg/d, up to a maximum of 80 mg/d for severe or resistant gastrointestinal disorders.11 Health Canada published safety advisories for domperidone in March 201212 and January 2015,13 urging caution with use and, in 2015, limiting doses for all indications to 30 mg/d. The initial 2012 warning was based on 2 observational studies that reported significant associations between domperidone use and ventricular arrhythmia and sudden cardiac death.14,15 However, because the study population included older patients (mean age in the 70s), many questioned the Health Canada warning and its applicability to a younger, relatively healthier patient population of breastfeeding women.16,17 Even less is known about the effect of domperidone on the nursing infant. The drug is detected at low levels in breast milk,11 and there are reports of QT prolongation with domperidone exposure at therapeutic levels in infants.18
The aim of this study was to examine trends in postpartum domperidone use in BC from 2002 to 2011. We examined the frequency of domperidone use over this time period, doses, duration and characteristics of women receiving domperidone prescriptions.
Methods
Data sources
Our analysis is based on de-identified, linked health data sets provided by Perinatal Services BC and Population Data BC with approval of relevant data stewards.19-22 All inferences, opinions and conclusions drawn in this publication are those of the authors, and do not reflect the opinions or policies of Perinatal Services BC or Population Data BC. Data sets included health care and sociodemographic information for all BC residents except those whose prescription drug coverage fell under federal jurisdiction (i.e., military veterans, registered First Nations people and Inuit, and federal penitentiary inmates, who collectively make up about 4% of the BC population).
Information about maternal and infant health came from Perinatal Services BC's Perinatal Data Registry. This province-wide database includes information on antenatal, intrapartum/delivery and postpartum maternal and infant care and outcomes for nearly all births in BC (about 99%). Some information on maternal reproductive history (e.g., previous miscarriages, stillbirths and premature deliveries) and health behaviours during pregnancy (e.g., smoking) is also recorded. The validity of the registry variables concerning maternal characteristics, newborn characteristics and delivery information that were used in this study is very high.23
The registry data were linked to information about women's prescription drug dispensations, medical services use, hospital admissions and income, as well as vital statistics for their babies. Information about domperidone dispensations came from PharmaNet, an information system that records every prescription filled at a pharmacy outside of acute care hospitals in BC, regardless of patient age or insurance status. Medical and hospital data were obtained from BC's universal, public health insurance program. Income quintiles were estimated using government records of the average adjusted household income in each patient's neighbourhood, with neighbourhoods including about 400-700 residents.24
Study population
We identified all women in the Perinatal Data Registry with a live birth between Jan. 1, 2002, and Dec. 31, 2011. To ensure accurate capture of domperidone dispensations and health information for all women in our cohort, we excluded women who did not reside in BC for at least 275 days in each year from 1 year preconception to 1 year postdelivery (n = 32 143).
We used the Aggregated Diagnosis Groups of the Johns Hopkins Adjusted Clinical Groups Case-Mix System (version 10.0) to identify women with chronic conditions based on diagnoses recorded in hospital and physician billing records. The case-mix system has been shown to be predictive of both drug use and expenditure in the BC population.25
Vital statistics data provided exact dates of birth for all babies born to the women in our cohort. For more than 90% of the study population, we were able to compute reliable gestational age estimates based on information from early-gestation ultrasonography, date of last menstrual period, infant clinical examination and/or chart documentation.
Outcome variables
Based on the date the prescription was filled, we characterized patterns of domperidone use using the following metrics: prevalence of use among women in the postpartum period; number of domperidone dispensations; daily dose per domperidone prescription; duration of individual prescriptions; timing of dispensation of the first prescription; and total days of domperidone therapy during the first 6 months postpartum. We examined all outcome variables by the type of birth (preterm or term). When there were clinically meaningful differences, we report the results separately; otherwise, we report pooled results.
To better understand the indication for which domperidone was prescribed, we examined the specialty of the prescribing physician and recorded diagnosis in the physician billing record for the visit that was associated with the first domperidone prescription. We were able to match 93% of prescriptions with physician specialty and 80% of prescriptions with records of related physician visits with information on diagnosis. Differences in the linkage rates stem from prescriptions received at hospital discharge or in non-fee-for-service medical clinics, for which there are no unique physician encounter data. To estimate use potentially associated with gastrointestinal motility disorders, we also examined the prevalence of prior diagnoses of disorders of the stomach (International Classification of Diseases, 9th Revision, code 536) during 1 year preconception and during pregnancy for all women dispensed domperidone during the postpartum period.
Oxytocin nasal spray and metoclopramide can also be used as galactagogues, but the former is not sold in Canada, and there were few postpartum metoclopramide prescriptions in our data set (< 100/yr). Therefore, we did not include any analyses on alternate galactogogues.
Statistical analyses
We present descriptive statistics for the metrics defined above. We used logistic regression to generate crude and adjusted odds ratios (ORs) and 95% confidence intervals (CIs) for health, demographic and socioeconomic maternal characteristics associated with postpartum domperidone use. We adjusted for clustering at the level of the mother because some women had given birth more than once during our study period. All analyses were performed using Stata version 13.1 and SAS version 9.3.
Ethics approval
This study was approved by the University of British Columbia's Behavioural Research Ethics Board (H10-01002; Apr. 19, 2010).
Results
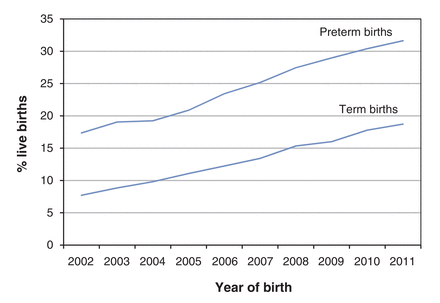
We identified 225 532 women who had 320 351 live births between Jan. 1, 2002, and Dec. 31, 2011. In 45 518 instances, women filled at least 1 domperidone prescription during the first 6 months of the postpartum period. The prevalence of postpartum domperidone use between 2002 and 2011 more than doubled, increasing gradually from 8% to 19% for term births and from 17% to 32% for preterm births (Figure 1). The mean (SD) and median (interquartile range [IQR]) time to first domperidone prescription postpartum increased, respectively, from 30 (SD 34) and 16 (IQR 9-37) days in 2002 to 35 (SD 40) and 18 (IQR 8-45) days in 2011 from the delivery date.
Domperidone use in the first 6 months postpartum among women in British Columbia with live births.
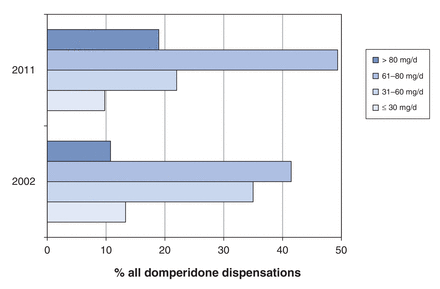
Prescribed doses rose over time in a similar fashion for both initial and subsequent prescriptions. Figure 2 shows the distribution of daily doses for all domperidone prescriptions in 2002 and 2011. For the initial prescriptions, the median daily dose increased from 60 (IQR 40-80) mg in 2002 to 80 (IQR 40-80) mg in 2011; for all prescriptions, it rose from 67 (IQR 40-80) mg to 80 (IQR 60-80) mg. In 2011, doses in the range of 61-80 mg/d made up half of all domperidone dispensations in that year, and dispensation for doses of greater than 80 mg/d almost doubled from 11% in 2002 to 19% in 2011. Of all subsequent prescriptions written for the study population in 2002, 32% of them were at a higher dose than initial prescriptions; for 2011, this figure was 26%, reflecting the higher starting dose of initial prescriptions in 2011.
Distribution of daily doses per dispensed domperidone prescription in 2002 and 2011.
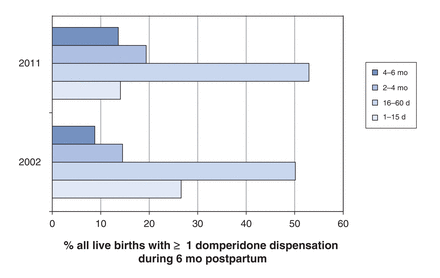
Individual prescription duration was longer in 2011 than in 2002: median duration was 21 (IQR 14-30) days in 2002 and 30 (IQR 15-30) days in 2011. The total number of days of domperidone therapy during the 6-month postpartum period also increased, from a mean of 47 to 59 days. In 2002, 8.8% of women with live births had prescriptions covering at least 120 of the first 180 days of the postpartum period, increasing to 14% in 2011. Conversely, the proportion with 15 or fewer days of use decreased from 27% in 2002 to 14% in 2011. Figure 3 illustrates the shift at the population level in both the number of women who took domperidone during the first 6 months postpartum and the duration of domperidone therapy per woman.
Duration of domperidone therapy during the first 6 months postpartum in 2002 and 2011.
Women who were older, had a higher body mass index, had a chronic disease, were first-time mothers, delivered more than 1 baby (multiple pregnancy), had a preterm birth or had a cesarian delivery were more likely to fill a domperidone prescription during the postpartum period (Table 1). There were no large differences by smoking status or by income quintile.
Prepregnancy prescriptions would be expected if women with gastrointestinal problems discontinued use in pregnancy and reinitiated postpartum. However, only 1.5% of women who used domperidone postpartum had a physician visit for disorders of the stomach during 1 year preconception or during pregnancy. Among first-time mothers who used domperidone postpartum, only 0.4% had at least 1 domperidone dispensation during 1 year preconception or during pregnancy.
The vast majority (92%) of both initial and subsequent domperidone prescriptions during the postpartum period were written by general practitioners, with almost all of the remaining 8% accounted for equally by pediatricians and obstetrician-gynecologists. The most frequent diagnoses recorded as the reason for physician visit associated with initial domperidone prescriptions are listed in Table 2. The top 2 diagnoses, accounting for 28% of matched records, were disorders of the breast. Less than 0.1% of domperidone dispensations were associated with a physician visit for disorders of the stomach.
Interpretation
Our study identifies an increase in the use of domperidone during the postpartum period among women in BC. Most initial domperidone prescriptions were written by general practitioners, and the most frequent indication for these prescriptions appears to be disorders of the breast.
There is very limited published information on prevalence and trends in postpartum domperidone use. Our findings are consistent with the results of a 2009 study involving a convenience sample of 76 postpartum women attending drop-in clinics in Calgary, Alberta, which found a 28% rate of domperidone use.26 Internationally, 1 report from an Australian hospital with outpatient postpartum services describes increased use over the same period, but at a much lower level: from less than 0.5% of women in 2000 to 5% in 2010.27 An analysis of prescribing patterns at this hospital indicates broadly similar patterns to those we observed in BC, including higher rates of domperidone use associated with preterm birth, cesarian delivery, primiparity, older maternal age and obesity.
The reasons for the increase in domperidone use postpartum in BC are likely multifactorial. The rate of exclusive breastfeeding for at least 6 months after birth in BC is the highest in Canada, and it has risen from 28% to 41% between 2003 and 2011/12.28 Campaigns directed at new mothers and clinicians during the 2000s about the health benefits of breastfeeding may have resulted in domperidone being seen as a solution in the face of potential lactation difficulties. There may have been an increase in demand for domperidone from patients. It is also possible that there has been a snowball effect in prescribing among physicians, with more relaxed prescribing practices becoming widespread in the absence of advisories against domperidone's use as a galactogogue. In Canada, domperidone is available only as a generic drug and is produced by 11 manufacturers. This makes manufacturer-driven promotion an unlikely contributor to off-label postpartum use.
In January 2015, Health Canada recommended domperidone use at no more than 30 mg/d, regardless of indication.13 The European Medicines Agency recommends a daily dose of 30 mg or less for no more than 1 week.29 This advice differs considerably from the doses and durations we observed in BC throughout the 2000s. It is possible that the observed dose escalation over the study period reflects inadequate efficacy (perceived or real) at lower doses. A patient information booklet accessed in April 2015 from the BC Women's Hospital lactation clinic, given to all mothers who have trouble breastfeeding, includes an extensive section on domperidone and recommends a daily dose of 30-80 mg for 3-8 weeks.30 Similarly, a recent patient information sheet from an Ontario breastfeeding clinic recommends an initial dose of 90 mg/d and duration of therapy for a minimum of 4-6 weeks.31
Limitations
Given our study design, we could not confirm whether participants were breastfeeding at the time of domperidone dispensation. Secondly, our study measured prescriptions filled and we have no way of tracking drug use. However, it should be noted that dispensation information is a more accurate measure of drug exposure than data on prescriptions written because, for a variety of reasons, many prescriptions written by doctors are not filled by patients.32 Further, there is no reason to believe that the proportion of filled prescriptions that are not taken would systematically differ over time.
Conclusion
We observed an increase in postpartum domperidone prescriptions from 2002 to 2011 as well as increased doses and duration of use. In 2011, 1 in 5 women was prescribed domperidone in the first 6 months after giving birth, with the majority of prescriptions above levels currently considered safe by regulators. Our results suggest routine - and increasing - postpartum use of domperidone in the face of uncertain safety to mothers and infants. More research is needed on maternal and infant health outcomes related to domperidone.
Supplemental information
For reviewer comments and the original submission of this manuscript, please see www.cmajopen.ca/content/4/1/E13/suppl/DC1.
Footnotes
Competing interests: None declared.
Contributors: All of the authors conceived the study design and approach. Steven Morgan acquired funding and data. Kate Smolina and Barbara Mintzes jointly planned the data analysis on postpartum domperidone use, and Kate Smolina undertook the analysis; Barbara Mintzes and Gillian Hanley carried out the evidence review. Kate Smolina drafted the paper. All of the authors reviewed results, contributed to interpretation and revised the manuscript. All of the authors gave final approval of the version to be published and agreed to act as guarantor of the work.
Funding: This study was supported by a grant from the Canadian Institutes of Health Research (CIHR DC0190GP). Kate Smolina is funded by a CIHR Banting Postdoctoral Fellowship.
References
- Copyright 2016, 8872147 Canada Inc.