Abstract
Background: Omicron is the current predominant variant of concern of SARS-CoV-2. We hypothesized that vaccination alters outcomes of patients hospitalized with COVID-19 during the Omicron wave and that these patients have different characteristics and outcomes than in previous waves.
Methods: This is a substudy of the Host Response Mediators in Coronavirus (COVID-19) Infection (ARBs CORONA I) trial, which included adults admitted to hospital with acute COVID-19 up to July 2022 from 9 hospitals in British Columbia, Ontario and Quebec. We excluded emergency department visits without hospital admission, readmissions and admissions for another reason. Using adjusted regression analysis, we compared mortality and organ dysfunction between vaccinated (≥ 2 doses) and unvaccinated patients during the Omicron wave, as well as between all patients in the Omicron and first 3 waves of the COVID-19 pandemic.
Results: During the Omicron wave, 28-day mortality was significantly lower in vaccinated (n = 19/237) than unvaccinated hospitalized patients (n = 12/127) (adjusted odds ratio [OR] 0.36, 95% confidence interval [CI] 0.15–0.89); vaccinated patients had lower risk of admission to the intensive care unit, invasive ventilation and acute respiratory distress syndrome and shorter hospital length of stay. Patients hospitalized during the Omicron wave had more comorbidities than in previous waves, and lower 28-day mortality than in waves 1 and 2 (adjusted OR 0.38, 95% CI 0.24–0.59; and 0.42, 95% CI 0.26–0.65) but not wave 3 (adjusted OR 0.81, 95% CI 0.43–1.51) and had less organ dysfunction than in the first 2 waves.
Interpretation: Patients who were at least double vaccinated had lower mortality than unvaccinated patients hospitalized during the Omicron wave. Patients hospitalized during the Omicron wave had more chronic disease and lower mortality than in the first 2 waves, but not wave 3. Changes in vaccination, treatments and predominant SARS-CoV-2 variant may have decreased mortality in patients hospitalized during the Omicron wave.
Omicron, the predominant variant of concern of SARS-CoV-2,1 is more transmissible but does not increase — and even decreases2,3 — risks of hospitalization4–7 and death8,9 compared with previous variants.
From March 2020 to July 2022, COVID-19 waves in Canada10 were driven by wild-type, Alpha, Delta and Omicron variants, and mortality decreased after wave 18,9,11–16 except in hospitalized patients aged 65 years or older in Ontario.17
Mortality of patients hospitalized during previous COVID-19 waves in Canada16 had decreased from waves 1 to 3 because of differing demographic characteristics, management (e.g., dexamethasone use, other COVID-19 therapies18,19) and vaccines.20 Overall, Omicron fatality rates decreased community wide.8,9
This study focuses on patients hospitalized with COVID-19 in 3 Canadian provinces (British Columbia, Ontario and Quebec) during the Omicron and first 3 waves. Our objectives were to compare outcomes of patients in the Omicron wave by vaccination status, and also to compare outcomes between patients hospitalized during the Omicron wave and patients in previous waves.
Methods
Study design
This substudy of the Host Response Mediators in Coronavirus (COVID-19) Infection (ARBs CORONA I) trial,21 a multicentre Canadian pragmatic observational cohort study examining pre-existing use of angiotensin receptor blockers and outcomes in patients hospitalized with COVID-19, is reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) checklist.22
Setting
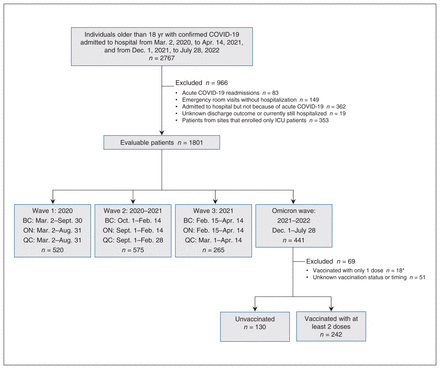
Sites that took part in ARBs CORONA I included community and teaching hospitals in BC, Ontario and Quebec (9 sites; Appendix 1, Table E1, available at www.cmajopen.ca/content/11/4/E672/suppl/DC1) that saw large numbers of patients hospitalized with acute COVID-19 (Mar. 2, 2020, to July 28, 2022). We chose these 3 provinces because they had a large number of COVID-19 patients and we could recruit sites for these provinces. Patients hospitalized during the Omicron wave were defined by admission to hospital for acute COVID-19 after Dec. 1, 2021, because Omicron had replaced Delta by then.23 Definitions of waves 1, 2 and 3 in BC, Ontario and Quebec were derived from the Canadian national COVID-19 daily epidemiology update website,24 as per our previous analysis16 (Figure 1). Patients were defined as being at least double vaccinated if they had received 2 or more doses of any type of SARS-CoV-2 vaccine (BNT162b2, mRNA-1273, ChAdOx1 and Ad26. COV2.S) before hospital admission (except for Ad26. COV2.S, for which a single vaccine dose would be considered double vaccinated). We used 2 vaccine doses to define “fully vaccinated” because that was the definition used by the Public Health Agency of Canada as of Sept. 1, 2022.25
Flow diagram showing patient selection. Note: BC = British Columbia, ICU = intensive care unit, ON = Ontario, QC = Quebec. *BC (n = 10/226; 4.4%) and ON (n = 8/184; 4.3%).
Participants
Inclusion criteria for ARBs CORONA I were people older than 18 years (regardless of pre-existing angiotensin receptor blockers usage) with SARS-CoV-2 infection confirmed by a clinically approved laboratory SARS-CoV-2 test from a local hospital or provincial laboratory, who were admitted to hospital for acute COVID-19, based on best evidence.26–30 Site investigators judged that the admitting illness was consistent with acute COVID-19. For the current study, we included all patients enrolled in ARBs CORONA I except for those with acute COVID-19 readmissions, emergency department visits without hospital admission and hospital admissions with positive SARS-CoV-2 test but whose illness was not acute COVID-19 (e.g., chest pain, appendicitis, fracture; Appendix 1, Table S1). We excluded sites that enrolled only patients admitted to the intensive care unit (ICU), as crude comparisons between waves would be confounded by the percentage of patients from these sites in each wave (Figure 1 and Appendix 1, Table S1).
Data sources
Patients were identified prospectively by research coordinators at each site, and data were collected on specifically designed electronic Case Report Forms (Appendix 2, available at www.cmajopen.ca/content/11/4/E672/suppl/DC1). Baseline data were available within 24 hours of admission. Booster vaccine dose data were not collected. Quebec sites did not recruit in wave 3, owing to research coordinator shortages.
Outcomes
The primary outcome was 28-day mortality; we assumed patients discharged alive before day 28 and lost to follow-up were day 28 survivors.19,31,32
Secondary outcomes included in-hospital mortality, ICU admission, organ dysfunction, hospital length of stay and concomitant treatments. We scored organ dysfunction first as frequency of septic shock, acute respiratory distress syndrome, acute kidney injury and acute cardiac injury, and second as frequency of the use of invasive mechanical ventilation, vasopressors and renal replacement therapy (RRT) and as days alive and free (DAF) of these therapies within the first 14 days.33 The DAF of ventilation, vasopressors and RRT calculation presents use of these therapies while adjusting for deaths in the first 14 days because many patients died within the first 14 days.26–30 Therefore, if we reported only duration of ventilation in a patient who died on day 2, the short duration of ventilation is biased by the early death. For patients who survived the first 14 days, DAF was simply the total number of days free from these therapies. Because mortality was a competing risk for the use of vasopressors, ventilation and RRT, patients who died within 14 days were assigned a DAF of 0 to increase the penalty for nonsurvival. We calculated DAF over 14 days, as in other trials in critically ill patients.34
Sample size
We performed no formal sample size calculation, as this was a substudy of ARBs CORONA I. The initial planned sample size of ARBs CORONA I was 49721 and was later changed to an open cohort because we obtained increased funding.
Statistical analyses
We compared baseline characteristics using the χ2 test, Fisher exact test (when > 20% of cells have expected cell counts < 5 or any expected cell count is < 1), analysis of variance (ANOVA) or Kruskal–Wallis test. We performed unadjusted and adjusted regression analyses to compare outcomes between vaccination groups, and between patients during the Omicron and previous waves.
To compare patients who were unvaccinated and patients with 2 or more vaccine doses in the Omicron wave, we adjusted for age, sex and comorbidities that were potentially associated with death, based on previous literature, and were significantly different between groups (chronic heart, kidney, hematologic diseases, hypertension and cancer). Patients with only 1 vaccine dose were not considered in the between-vaccination group comparisons, given the limited number.
For wave comparisons, we adjusted for predefined factors: age, sex, comorbidities (chronic heart disease, hypertension, chronic kidney disease and diabetes), commonest comorbidities associated with death,35–37 baseline systolic blood pressure, organ dysfunction confounders that were different across waves (baseline heart rate, arterial oxygen saturation [Sao2] and serum creatinine), and comorbidities that were different across waves (chronic pulmonary disease, chronic neurologic disorder, cancer and chronic hematologic disease). We accounted for site effect in unadjusted and adjusted comparisons between waves by including a hospital site effect term in the regression model, as the regional distribution of patients was different across waves, owing to the varying levels of site participation over time.
Given the smaller sample size for comparisons within the Omicron wave by vaccination status, we used a different strategy of adjustment variables selection than the wave comparisons to reduce the possibility of overfitting.
We used logistic and censored quantile38 regression to compare binary outcomes and length of stay, respectively. The observed DAF data exhibited a U-shape distribution, with most data concentrated at 0 and 14. We thus used 0–1 inflated β regression39 to model the data.32,40 Days alive and free were expressed as proportion of days (i.e., divided by 14) in the regression model and then back-transformed for interpretation. The R package gamlss was used to fit the 0–1 inflated β model with the default logit and log link functions. Given that the regression model was adjusted for covariates, we computed the marginal mean DAF by vaccination status and wave by averaging the model predictions obtained from the predict function (i.e., fixed vaccination status/wave at a specific value and integrating over the remaining covariates). We obtained estimated mean difference between vaccination statuses and waves by taking the difference between the marginal means. We obtained 95% confidence intervals (CIs) using 1000 bootstrap samples, and obtained p values for the comparisons by resampling under the null hypothesis of no difference (1000 samples).
For comparison within the Omicron wave by vaccination status, we applied Firth’s penalized method to logistic regression because of the number of adjustment factors and low event count for some outcomes. For wave comparisons, we considered hospital site as a random effect in logistic regression and as fixed in censored quantile and 0–1 inflated β regression, owing to numerical issues and computational limitations. Results were expressed as odds ratio (OR), mean difference in DAF and difference in median length of stay (ΔM) with 95% CIs. For length-of-stay analysis, we considered in-hospital deaths as never discharged and censored them at the largest observed length of stay.41
Missing data were minimal, so we excluded patients with missing data from the corresponding analysis (about 5% for the adjusted analysis). We conducted analyses using SAS 9.4 (SAS Institute Inc.) and R 4.0.4 (R Foundation for Statistical Computing). We considered p < 0.05 to be statistically significant without adjustment for multiple testing.
Ethics approval
This study was approved by Providence Health Care and University of British Columbia Human Research Committee and by each of the contributing clinical sites. Anonymized clinical data were deemed low risk and informed consent was not required.
Results
Comparison by vaccination status within the Omicron wave
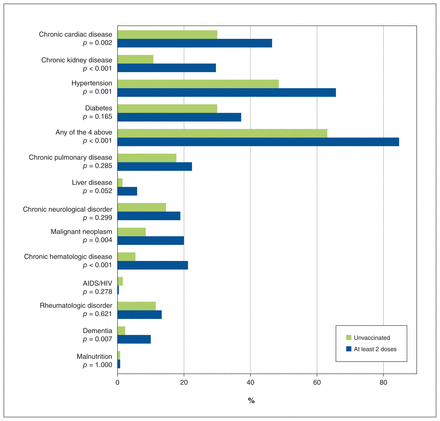
More than half of patients hospitalized during the Omicron wave had had 2 or more doses of SARS-CoV-2 vaccine before hospital admission (242/390 [62%]; 92% were mRNA vaccines [222/242]; all had received the second dose > 14 days before admission). Patients with 2 or more vaccine doses were older and had more frequent comorbidities (chronic cardiac, kidney and hematologic diseases, hypertension, cancer and dementia) but less ICU admission on hospital admission day (Table 1; Appendix 1, Table E2; and Figure 2). Treatments during hospital stay were similar, except for less dexamethasone use in the group with 2 or more vaccine doses (65.7% v. 76.2%, p = 0.04; Appendix 1, Table E3).
Baseline characteristics of patients admitted to hospital during the Omicron wave with acute COVID-19, by SARS-CoV-2 vaccination status
Comorbidities of patients hospitalized during the Omicron wave, by vaccination group. P value based on χ2 test or Fisher exact test, as appropriate.
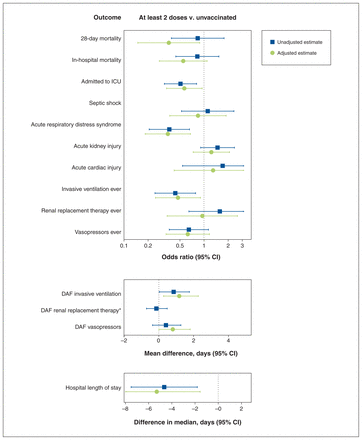
Vaccine appeared protective. Crude 28-day mortality was slightly lower in the group with 2 or more doses than in the unvaccinated group (8.0% v. 9.4%; OR 0.84, 95% CI 0.39–1.78; Table 2); in the adjusted comparison, mortality was significantly lower for the group with 2 or more doses (adjusted OR 0.36, 95% CI 0.15–0.89; Figure 3). Admission to the ICU, invasive ventilation and acute respiratory distress syndrome were less likely in the group with 2 or more doses group (respectively: adjusted OR 0.57, 95% CI 0.34–0.95; 0.47, 95% CI 0.25–0.90; and 0.35, 95% CI 0.18–0.68). Hospital length of stay was significantly shorter in the group with 2 or more doses (adjusted ΔM −5.3 d, 95% CI −9.1 to −1.6) (Figure 3).
Outcomes of patients admitted to hospital for acute COVID-19 during the Omicron wave by SARS-CoV-2 vaccination status
Comparison of outcomes of hospitalized patients with ≥ 2 doses of SARS-CoV-2 vaccine versus no vaccine during the Omicron wave, by regression analysis. Note: CI = confidence interval, DAF = days alive and free, ICU = intensive care unit. Note: The following factors were accounted for in the adjusted analysis: age, sex, chronic heart disease, chronic kidney disease, hematologic disease, hypertension and cancer. *Adjusted regression analysis was not feasible numerically as too few patients received renal replacement therapy during the first 14 days.
Comparison of Omicron wave with previous waves
Baseline characteristics
Patients hospitalized during the Omicron wave (n = 441) had more frequent comorbidities (chronic cardiac, kidney, pulmonary, neurologic and hematologic diseases; hypertension; and cancer) than patients hospitalized during previous waves (Appendix 1, Table E4 and Figure E1). Vaccination proportions were 0%, 0.5%, 8.2% and 66.8% in waves 1, 2, 3 and Omicron, respectively (p < 0.001). D-dimer levels were elevated in patients hospitalized during the Omicron wave versus during previous waves.
Treatment
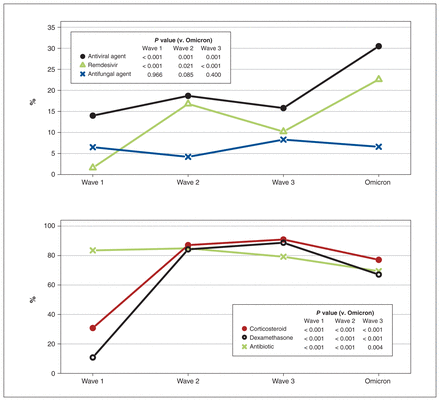
Patients admitted during the Omicron wave had more remdesivir use (22.6%) than those in previous waves (wave 1: 1.6%, p < 0.001; wave 2: 16.8%, p = 0.02; wave 3: 10.2%, p < 0.001), but less dexamethasone use (67.1%) than patients during wave 2 (84.2%, p < 0.001) and wave 3 (88.7%, p < 0.001) (Figure 4 and Appendix 1, Table E5).
COVID-19 therapies administered during patients’ hospital stay across COVID-19 waves. p value was based on χ2 test or Fisher exact test, as appropriate.
Outcomes
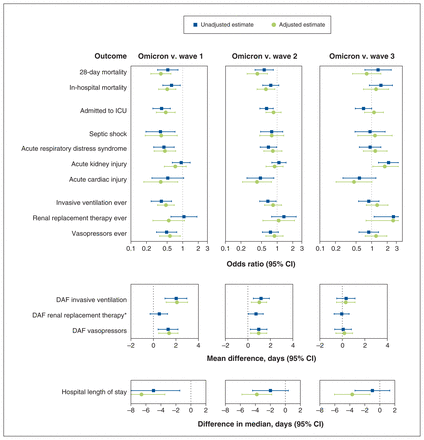
The 28-day mortality of patients hospitalized during the Omicron wave was significantly lower than in the first 2 waves (adjusted OR 0.38, 95% CI 0.24–0.59; and 0.42, 95% CI 0.26–0.65 v. waves 1 and 2, respectively), but not wave 3 (adjusted OR 0.81, 95% CI 0.43–1.51). During the Omicron wave, patients admitted to hospital had less organ dysfunction than those admitted in waves 1 and 2, but not in wave 3 (Figure 5 and Appendix 1, Table E6). During the Omicron wave, patients admitted to hospital had significantly more acute kidney injury than those in wave 3 (adjusted OR 1.80, 95% CI 1.06–3.05). Hospital stay was shorter during Omicron than all 3 earlier waves.
Comparison of outcomes of patients hospitalized with COVID-19 during the Omicron versus previous waves, by regression analysis. Note: The following factors were accounted for in the adjusted analysis: age, sex, comorbidities (chronic heart disease, hypertension, chronic kidney disease, diabetes, chronic pulmonary disease, chronic neurologic disorder, cancer and chronic hematologic disease), baseline systolic blood pressure, heart rate, oxygen saturation of arterial blood (Sao2 ) and serum creatinine. Note: CI = confidence interval, DAF = days alive and free, ICU = intensive care unit. *Adjusted regression analysis was not feasible numerically as too few patients received renal replacement therapy during the first 14 days.
The magnitude of the wave effect on 28-day mortality in the supplementary analysis restricted to unvaccinated patients (Appendix 1, Figure E2) was slightly smaller (adjusted OR 0.47, 95% CI 0.23–0.96; 0.53, 95% CI 0.26–1.08; and 1.00, 95% CI 0.42–2.36 for the Omicron wave than waves 1, 2 and 3, respectively), and with much wider CIs, as the sample size for the Omicron wave was reduced by two-thirds.
Interpretation
Patients hospitalized during the Omicron wave with at least 2 doses of SARS-CoV-2 vaccine had significantly more comorbidities than non-vaccinated patients admitted at that time. Vaccination mitigates hospitalization risk,5,42–46 but it could be less effective in patients with comorbidities. 44,47 Thus, we expected that hospitalization among vaccinated patients would be concentrated in those with many comorbidities, and that was what we observed. Mortality (and ICU admission, invasive ventilation and acute respiratory distress syndrome) of the vaccinated patients was significantly lower than in unvaccinated patients after adjustment for confounders.
The significantly higher rates of chronic kidney, pulmonary and hematologic disease, and malignancy of patients hospitalized during the Omicron wave suggested evidence of more underlying immunosuppression, which could increase risk of hospitalization.44,48
Patients admitted during the Omicron wave had more frequent vaccination before hospitalization than in previous waves, as SARS-CoV-2 vaccines were not previously available. Vaccines were rolled out in various stages in each province, so the proportions of vaccinated patients would be expected to be lower at the beginning of the pandemic. Indeed, we found that the proportions of vaccinated patients were 0%, 0.5%, 8.2% and 66.8% in waves 1, 2, 3 and Omicron, respectively. The proportions of vaccinated patients in each wave probably do not explain the lack of differences in mortality between waves 3 and Omicron because of the large differences in vaccination rates but similar mortality rates (Appendix 1, Tables E4–E6).
Double vaccination appears somewhat less effective against infection with the Omicron variant than earlier variants in mitigating hospitalization and death rates.5,42–46 However, SARS-CoV-2 vaccination immunity may wane, increasing later risk of acute COVID-19 illness severity. 5,43,44,46 Booster vaccination increases immunity49 and decreases severe infection risk during the Omicron wave.50–52 A fourth dose of SARS-CoV-2 vaccine53 and previous SARS-CoV-2 infection plus boosters were most protective. More frequent vaccination during the Omicron wave than in previous waves may have affected the patient mix.5,46
Patients hospitalized during the Omicron wave had more remdesivir use than in previous waves but less dexamethasone use than in waves 2 and 3. They had lower 28-day mortality and less organ dysfunction than patients from waves 1 and 2, but not wave 3. However, there appeared to be a higher rate of acute kidney injury in patients hospitalized during the Omicron wave than in wave 3. A study from Italy with 65 critically ill patients found that the Omicron variant was associated with more acute kidney injury than the Delta variant.54
Severe COVID-19 may be associated with a procoagulant state and greater risk of venous thromboembolic disease.55,56 We found that D-dimer levels were very commonly elevated in patients hospitalized during the Omicron wave: a procoagulant state may be as important in these patients as in those hospitalized during previous waves.
Omicron is more transmissible but has lower risk of hospital admission and death than previous variants of concern.7,15,57,58 Our study sheds new light on the Omicron wave: the risk of hospitalization may be lower with Omicron than previous variants of concern, and once these patients are hospitalized, the mortality rate is lower (about 10%) than in patients during the first 2 waves. This could be the result of changes in vaccination, treatments and predominant SARS-CoV-2 variant.
Other studies outside Canada that compared Omicron with other variants also found lower rates of ICU admission and mortality in hospitalized patients.2,3,5,59 We extend those reports and our previous study16 by showing lower mortality during the Omicron wave than in waves 1 and 2, but similar mortality to wave 3 in Canada, and highlight specific organ dysfunction and support needs during the Omicron wave.
Our study strengths include the multicentre open cohort design, the detailed phenotyping of inpatients, and the large control group of patients from previous COVID-19 waves in Canada.
Limitations
In this observational study, we could not determine causation, but add evidence regarding differences in characteristics, treatments and outcomes between vaccination groups, as well as COVID-19 waves. The sample size for the comparison by vaccination status within the Omicron wave was limited and the number of events for some outcomes were low. This may limit statistical power and reliability of the adjusted analysis. We also performed a large number of hypothesis tests without adjustment for multiplicity. We did not collect data on booster vaccine doses and so cannot determine the timing between the last vaccine dose and hospital admission and, thus, are unable to tease out their impact on outcomes. Comorbidities were identified at time of admission, but the duration was not recorded; nor was treatment and level of control for these comorbidities, and these comorbidity variables may have affected mortality risk. A more effective risk adjustment on mortality would have been achieved by using the Charlson or Elixhauser comorbidity indices. However, we did not collect several of the variables that are used in each index and so cannot report a post hoc calculation of either index.
We did not capture use of other COVID-19 therapies — in particular, anticoagulants and immunomodulatory drugs — which could alter patient outcomes. We did not assess neurologic function because neurologic dysfunction assessment in patients who are critically ill is difficult, owing to the confounding effects of sedation. Hospital and ICU admission for COVID-19 is primarily for respiratory, cardiovascular and renal support; we measured use of ventilation, vasopressors and RRT and calculated DAF of such support. 16,33 We did not adjudicate whether patients had acute COVID-19; however, we engaged large centres with extensive experience of acute COVID-19 in BC, Ontario and Quebec. Having only 3 provinces somewhat limits the representativeness of our study.
Further limitations are that we did not have variants of concern determined for individual patients, but the national data during the time frame we defined as the Omicron wave demonstrated that more than 90% of cases of SARS-CoV-2 infection were the Omicron variant.24 We confirmed by genome sequencing that 94% of patients during the Omicron wave were infected with Omicron, in a BC subset (n = 141; December 2021 to April 2022). However, this may not rule out overlapping variants. Our research coordinators used SARS-CoV-2–positive tests in the hospital laboratory to identify patients, but some patients may have been missed.
Conclusion
At-least double-vaccinated patients had lower mortality than unvaccinated patients hospitalized with COVID-19 during the Omicron wave. These patients had more chronic disease and lower mortality than those admitted in waves 1 and 2, but not in wave 3. Changes in vaccination, treatments and predominant SARS-CoV-2 variant may have decreased mortality in patients hospitalized with COVID-19 during the Omicron wave.
Footnotes
Competing interests: Keith Walley reports receiving a Canadian Institutes of Health Research (CIHR) Foundation grant, paid to the University of British Columbia. Dr. Walley has also participated on data safety monitoring boards (unpaid) for Northern Therapeutics and the Cellular Immuno-Therapy for COVID-19 Acute Respiratory Distress Syndrome (CIRCA-19) trial. Allison McGeer reports receiving grants from Sanofi, Merck and Pfizer (paid to institution), as well as payment or honoraria from AstraZeneca, Merck, Biogen and Moderna. Dr. McGeer has also received travel support from Moderna and has participated on a data safety monitoring or advisory board for Pfizer, GlaxoSmithKline, Moderna, Medicago, Janssen, AstraZeneca, Novavax and Sanofi.
This article has been peer reviewed.
Contributors: Terry Lee, Joel Singer, Karen Tran, Puneet Mann, Kathryn Donohoe, Geraldine Hernandez and James Russell contributed to the conception and design of the work. Terry Lee contributed to the analysis of the data. All of the authors contributed to the interpretation of the data, drafted the manuscript, revised it critically for important intellectual content, gave final approval of the version to be published and agreed to be accountable for all aspects of the work.
Funding: This study is funded by grants from the Canadian Institutes of Health Research (CIHR #439993) to James A. Russell and St. Paul’s Foundation to James A. Russell.
Data sharing: The data are not available for use by other researchers.
Supplemental information: For reviewer comments and the original submission of this manuscript, please see www.cmajopen.ca/content/11/4/E672/suppl/DC1.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/
References
- © 2023 CMA Impact Inc. or its licensors