Abstract
Background Obesity is a major public health issue. This review updates the evidence on the effectiveness of behavioural and pharmacologic treatments for overweight and obesity in adults.
Methods We updated the search conducted in a previous review. Randomized trials of primary-care-relevant behavioural (diet, exercise and lifestyle) and pharmacologic (orlistat and metformin) with or without behavioural treatments in overweight and obese adults were included if 12-month, postbaseline data were provided for weight outcomes. Studies reporting harms were included regardless of design. Data were extracted and pooled wherever possible for 5 weight outcomes, 6 secondary health outcomes and 4 adverse events categories.
Results We identified 68 studies, most consisted of short-term (≤ 12 mo) treatments using diet (n = 8), exercise (n = 4), diet and exercise (n = 10), lifestyle (n = 19), orlistat (n = 25) or metformin (n = 4). Compared with the control groups, intervention participants had a greater weight loss of −3.02 kg (95% confidence interval [CI] −3.52 to −2.52), a greater reduction in waist circumference of −2.78 cm (95% CI −3.34 to −2.22) and a greater reduction in body mass index of −1.11 kg/m2 (95% CI −1.39 to −0.84). The relative risk for loss of ≥ 5% body weight was 1.77 (95% CI 1.58–1.99, [number needed to treat 5, 95% CI 4–7]), and the relative risk for loss of ≥ 10% body weight was 1.91 (95% CI 1.69–2.16, [number needed to treat 9, 95% CI 7–12]). Incidence of type 2 diabetes was lower among pre-diabetic intervention participants (relative risk 0.62 [95% CI 0.50–0.77], number needed to treat 17 [95% CI 13–29]). With prevalence rates for type 2 diabetes on the rise, weight loss coupled with a reduction in the incidence of type 2 diabetes could potentially have a significant benefit on population health and a possible reduction in need for drug treatments for glycemic control.
Interpretation There is moderate quality evidence that behavioural and pharmacologic plus behvioural, treatments for overweight and obesity in adults lead to clinically important reductions in weight and incidence of type 2 diabetes in pre-diabetic populations. Registration: PROSPERO no. CRD42012002753
Overweight and obesity are defined by a body mass index (BMI) of 25–29.9 and ≥ 30 kg/m2, respectively. An estimated one billion adults are overweight and at least 300 million are obese worldwide, with prevalence increasing in most countries.1 Obese adults are at increased risk for developing major diseases, such as type 2 diabetes, coronary artery disease, stroke, depression and certain cancers.2–4 It is estimated that one in 10 premature adult deaths is directly attributable to overweight and obesity.5,6
We provide an updated synthesis of the effectiveness of behavioural and pharmacologic interventions for treating overweight and obesity in adults. Whereas most systematic reviews on this topic have focused on anthropometric (e.g., weight and waist circumference)7,8 and biochemical outcomes (e.g., cholesterol and blood pressure),9–11 we aimed to also assess the effects of nonsurgical weight-loss interventions on clinically meaningful outcomes (e.g., 5% weight loss and incidence of type 2 diabetes).
Methods
Search strategy
A recent high-quality (9/11 AMSTAR rating)12 review by the United States Preventive Services Task Force13 examined interventions for preventing obesity in overweight and obese populations. To avoid duplication, our protocol was designed to update their search. We searched MEDLINE, Cochrane Central Register of Controlled Trials, PsycINFO and Embase from September 2010 (the date of the last United States Preventive Services Task Force search) up to and including April 19, 2013. The full search strategy is provided in Appendix 1 (available at www.cmajopen.ca/content/2/4/E306/suppl/DC1). Reference lists from other systematic reviews were searched for studies not captured by our search.
Population, intervention, comparator, outcome and setting statement
Details regarding the population, intervention, comparator, outcomes and setting for this review are provided in Box 1.
Description of population, intervention, comparator, outcomes and setting
Population
• Overweight (BMI 25–29.9 kg/m2) and obese (BMI 30–39.9 kg/m2) adults aged 18 years and older
Interventions
• Behavioural (diet, exercise or lifestyle) and pharmacologic (orlistat or metformin) treatments for weight loss
Comparator
• Treatment effectiveness: no intervention, usual care, placebo or minimal intervention (e.g., newsletter or single information session on healthy living)
• Treatment harms: any type of comparison group or no comparison group
Outcomes
• Treatment effectiveness: Primary weight outcomes: weight change in kg, loss of ≥ 5% and ≥ 10% baseline body weight, change in BMI, change in waist circumference. Secondary health outcomes: total cholesterol, low density lipoprotein cholesterol, fasting blood glucose, incidence of type 2 diabetes, and systolic and diastolic blood pressure
• Treatment harms: Any adverse events, serious adverse events (requiring admission to hospital or urgent medical care), gastrointestinal events, and withdrawal from the study because of adverse events
Settings
• Generalizable to Canadian primary care or feasible for conducting in or referral from primary care; surgical and metabolic unit interventions were excluded as representing a level of obesity and comorbid conditions that would be less commonly used as referral points from primary care
Inclusion and exclusion criteria
The inclusion and exclusion criteria for this review are provided in Box 2.
Inclusion and exclusion criteria
Studies were included if they met the following criteria:
• Behavioural (diet, exercise or lifestyle strategies), pharmacologic (orlistat or metformin) or combined strategy trial of weight loss treatment or management
• Intervention focused on adults ≥ 18 years old who were overweight (BMI 25–29.9 kg/m2) or obese (BMI 30–39.9 kg/m2)
• Randomized controlled trial with a no-intervention, usual care, placebo or minimal component (e.g., single newsletter or information session on general health) comparison group (this condition applied only to studies assessing treatment effectiveness)
• Reported data for one or more specified weight outcomes (i.e., weight change in kg, loss of ≥ 5% and ≥ 10% of baseline body weight, change in waist circumference, and change in BMI)
• Reported data for outcomes of interest at least 12 months postbaseline assessment
• No restrictions on study design, comparison group, reporting of weight outcome or timing of assessment were applied to studies that reported data for harms of treatment
• Results were published in English or French
Studies were excluded if:
• Treatment involved a surgical intervention or a drug other than orlistat or metformin
• Intervention focused on morbidly obese adults (BMI ≥ 40 kg/m2) or specifically enrolled participants who were pregnant, had an eating disorder or a condition in which weight gain was a cardinal manifestation (e.g., metabolic syndrome or polycystic ovarian disease).
• Intervention was conducted in an inpatient hospital, institutional or occupational setting or involved a school-based or faith-based program
• The only available results were published in a language other than English or French
Study selection, quality assessment and data abstraction
Titles and abstracts of papers were reviewed independently by 2 team members. Any citation marked for inclusion by either team member went on to full-text screening, which was also done independently by 2 researchers. Randomized controlled trials were assessed using the Cochrane Collaboration’s tool for risk-of-bias assessment14 (Appendix 2, available at www.cmajopen.ca/content/2/4/E306/suppl/DC1). Overall strength of the evidence (identified as high, moderate, low or very low quality) was determined using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) framework (GRADEpro version 3.2).15 One team member completed full-data abstraction and a second member verified all extractions. All data were checked in a third round of verification before analysis. Unadjusted immediate postassessment data were extracted where available. For a small number of studies, there were no immediate postassessment data; we chose the data point closest to the end of the intervention. For another small group of on-going studies, we extracted interim data (at 12 mo). We extracted data for all reported adverse events. At all levels, inter-rater disagreements were resolved through discussion.
Data analysis
For meta-analyses, immediate post-treatment data (means and standard deviations) were used for continous outcomes such as weight in kg, whereas number-of-events data were used for binary outcomes such as loss of ≥ 5% baseline body weight. The DerSimonian and Laird random-effects model with an inverse variance method was used to generate the summary measures of effect in the form of mean difference for continuous outcomes and risk ratio (RR) for binary outcomes.16 For studies with more than one treatment arm, we took different approaches depending on the similarity of the interventions. For similar interventions (e.g., 2 arms of a lifestyle intervention, one using phone contact and one using in-person support) we pooled the data to do a pairwise comparison with the control group. Alternatively, if groups were substantially different (e.g., low-calorie diet and high-intensity aerobic exercise) we included the data for each arm compared with the control group but split the sample size for the control group to avoid a unit-of-analysis error and double counting.14 All orlistat studies included an intervention of 120 mg thrice daily. Some studies also assessed smaller doses, but we only extracted data for effects of the consistently reported 120 mg dosage. Cochran’s Q (α = 0.10) and I2 statistics were used to quantify heterogeneity within and between subgroups. Sensitivity analyses were performed to evaluate statistical stability and effect on statistical heterogeneity. For the outcome of weight in kg, we did subgroup analyses by focus of intervention (behavioural and pharmacologic plus behavioural) for all outcomes and comparisons (except sex for weight in kg and gastrointestinal adverse effects, which included studies in a single group).
Results
The search and selection process is presented in Figure 1. Sixty-eight studies (117 papers) were eligible for inclusion in this review.17–84 Thirty-six of these studies were brought forward from the 2011 USPSTF review13 that met our inclusion criteria and 32 studies were found in the recent literature. Of the 68 studies, 54 randomized controlled trials reported weight outcome data that could be pooled; 2 others provided eligible weight data that could not be pooled.51,55 The remaining 12 studies (2 studies were single-group pre-post designs, 1 study had a more active comparison group and 9 studies reported outcomes at < 12 mo) were only included in analyses of adverse events.24,29,57,63,75–81,83 High within-group heterogeneity was common; however, the direction of treatment effect was consistent across most studies and the confidence intervals overlapped. This statistical heterogeneity is likely due to small versus large treatment effects observed across studies. Table 1 presents a summary of the features of this body of evidence; details for individual studies are provided in the Appendix 3 (available at www.cmajopen.ca/content/2/4/E306/suppl/DC1).
Search and selection flow diagram for articles on treatment of overweight and obesity in adults.
Weight outcomes
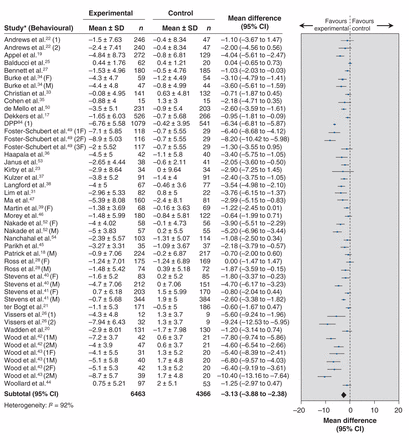
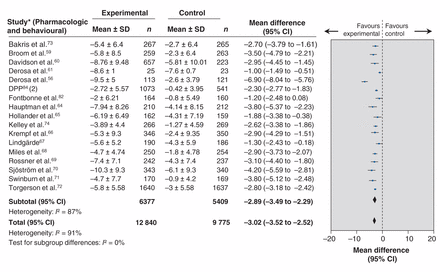
Forty-nine studies were included in the meta-analysis assessing weight change in kg.17–23,25–28,31,33–47, 49,50,52–54,56,59–61,64–74,82,84 Intervention participants had a significantly greater reduction in weight compared with the control group (Table 2 and Figures 2A and 2B). There was no evidence that the effect of treatment differed based on focus of intervention (behavioural or pharmacologic plus behavioural). Weight loss was greater in intervention participants than in control participants for both behavioural interventions and pharmacologic plus behavioural interventions. The test for subgroup differences based on type of behavioural intervention in the behavioural trials (diet, exercise, diet + exercise and lifestyle) was significant: interventions using exercise alone did not lead to significantly greater reductions in weight, whereas diet alone showed the largest difference between groups (Table 2). In the behavioural trials, the test for subgroup differences based on cardiovascular disease risk status was also significant: compared with the control group, changes in weight were greater for participants with low baseline risk than those with high baseline risk (Table 2). There was no evidence that the effect of treatment differed based on any of the other variables considered in the subgroup tests (i.e., intervention duration, sex, type of drug and cardiovascular disease risk in pharmacologic trials) (Table 2).
Effect of behavioural treatment interventions on weight in kilograms. Note: 1 = intervention arm 1; 2 = intervention arm 2; F = females only; M = males only: 1F, 2F and 3F represent female participants in different intervention arms; 1M and 2M represent male participants in different intervention arms; CI = confidence interval; DPP = Diabetes Prevention Program; SD = standard deviation.
Effect of pharmacologic plus behavioural treatment interventions on weight in kilograms. Note: 2 = intervention arm 2; CI = confidence interval; DPP = Diabetes Prevention Program; SD = standard deviation.
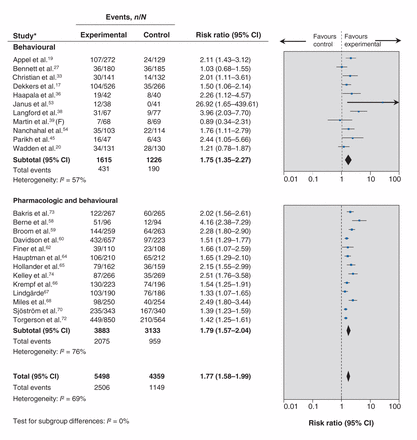
Twenty-four studies were included in the meta-analysis assessing loss of ≥ 5% baseline body weight.17,19,20,27,33,36,38,39,45,53,54,58–60,62,64–68,70,72–74 Figure 3 shows intervention participants were more likely to lose ≥ 5% of their baseline body weight compared with control participants. There was no evidence that the effect of treatment differed based on focus of intervention (Table 3).
Effect of treatment interventions on loss of ≥ 5% baseline body weight: overall and by focus of intervention (behavioural and pharmacologic plus behavioural). Note: CI = confidence interval; F = females only.
Similarly, 16 studies were included in the meta-analysis assessing loss of ≥ 10% baseline body weight.19,20,36,58–60,62,64–70,72,74 Intervention participants were more likely to lose ≥ 10% of their baseline body weight compared with controls. There was no evidence that the effect of treatment differed based on focus of intervention (Table 3).
Twenty-six studies were included in the meta-analysis assessing change in BMI from baseline.18–23,25–28,30,32,37,46–50,52–54,56,61,66,73,84 Intervention participants had a greater reduction in BMI compared with the control groups. There was no evidence that the effect of treatment differed based on focus of intervention (Table 4).
Thirty-three studies were included in the meta-analysis assessing change in waist circumference from baseline.17–22,25,26,28,33,34,36,37,45–47,49,50,52–54,56,58,59,61,65,66,69,71–74,84 Intervention participants had a greater reduction in waist circumference compared with controls. There was no evidence that the effect of treatment differed based on focus of intervention (Table 4).
Secondary outcomes
Meta-analyses showed greater improvements in intervention participants compared with control participants across all continuous secondary outcomes: total cholesterol,17,19–23,25,28,31,33,37,42,43,46–48,50,53,56,58,59,61,62,64,65,67–71,73,74,82 low-density lipoprotein cholesterol,19–23,25,28,31,33,42,43,45–47,53,56,58, 59,61,62,64,65,67–71,73,74,82 fasting blood glucose,19–22,26,28,31,37,45–48,50,53,56,58,64–72,74,82,84 and systolic17,19–22,25–28,31–34,37,40,42,43,45,47,53,54,58–61,64,66–74,82,84 and diastolic17,19–22,25–28,31–34,37,40,42,43,45,47,53,54,58–61,64,66,67,69–74,82,84 blood pressure (Table 4). For all but 2 of these outcomes, there was no evidence that the effect of treatment differed based on focus of intervention. The tests for subgroup differences were significant for total cholesterol and fasting glucose (Table 4). In both cases, when compared with the control groups, benefits were greater for participants in pharmacologic plus behavioural interventions than for those taking part in behavioural interventions alone.
Nine studies were included in a meta-analysis assessing the risk of type 2 diabetes in prediabetic patients.45–47,50,51,55,72,82,84 A diagnosis of new onset type 2 diabetes was less likely to occur in intervention participants compared with the control group (Table 3). There was no evidence that the effect of treatment differed based on focus of intervention (Table 3).
Adverse effects
Very few behavioural studies reported adverse events. When they did, harms were usually minor and related to injuries sustained during physical activity (e.g., joint, back or muscle pain; minor abrasions, bruises or blisters; and fractures). Most (about 80%) adverse events that occurred in orlistat trials (and some in metformin trials) were gastrointestinal disturbances. Commonly reported symptoms across studies were fatty or oily stool, increased defecation, increased urgency, abdominal pain, soft stools, oily spotting and flatulence. Most studies reported that the gastrointestinal events were typically mild or moderate in intensity and occurred only once or twice in the participants, usually near the beginning of treatment.
Across the 17 studies with data that could be pooled, intervention participants were more likely to have an adverse event compared with control participants (Table 5).58–61,64,66,67,69–74,82,84 However, as indicated by the test for subgroup differences, participants in the 15 pharmacologic interventions were significantly more likely to have an adverse event (Table 5). Serious adverse events were defined as those requiring urgent medical care or admission to hospital, as well as those defined as serious by the primary authors. A meta-analysis of 14 studies showed no difference between the intervention and control groups for the risk of having serious adverse events, and there was no evidence that the effect of treatment differed based on focus of intervention (Table 5).20,27,57,59,61,66,67,70–73,77,79,84 Gastrointestinal events were reported only in studies that used drug interventions and in the 23 studies with data that could be pooled; those taking active medications were more likely to report these events than control participants.57,59,61–69,71–77,79,81–84 Likewise, participants in 25 pharmacologic studies were more likely to withdraw from their study because of adverse events compared with control participants (Table 5).46,56,58–77,79,81,82
Interpretation
Main findings
There are 3 principal findings from this review. First, the pooled-effect estimates for all weight outcomes were statistically significant in favour of the interventions and, compared with the control groups, intervention participants had, on average, a 3.02 kg greater weight loss, a 2.78 cm greater reduction in waist circumference, and a 1.11 kg/m2 greater reduction in BMI, and were more likely to lose ≥ 5% (RR 1.77) and ≥ 10% (RR 1.91) of their baseline body weight. Every kilogram of weight loss in people with impaired glucose tolerance is associated with a 16% reduction in the incidence of type 2 diabetes.85 Second, there was no significant difference between behavioural and pharmacologic interventions for any weight outcomes, although the potential for adverse outcomes appears greater with pharmacologic treatments. Third, modest weight reduction, corresponding to loss of ≥ 5% and ≥ 10% of baseline body weight (number needed to treat 5 and 9, respectively) had clinically important effects, most notably a 38% reduction in the incidence of type 2 diabetes in prediabetic populations (number needed to treat 17). With prevalence rates for type 2 diabetes in the US and the European Union of 9.3% and 10%, respectively,86 coupled with its increasing prevalence, extrapolation of a 38% reduction in the incidence of type 2 diabetes could have a significant benefit on population health. Intervention participants also had small improvements in secondary outcomes; these effects may be of minor clinical significance at the individual level, but important at the population level.
The benefits of treatment should be weighed against the harms. Few behavioural studies reported adverse events; those that did found a small absolute excess in the risk of injuries associated with physical activity. Participants on active medications reported significantly milder to moderate gastrointestinal disturbances than those on placebo.
Comparison with other studies
Updating the United States Preventive Services Task Force review search13 added 32 studies, but did not point to changes in any important outcomes. We used a more comprehensive approach of subgrouping studies based on behavioural interventions such diet, exercise and lifestyle changes to better reflect the evidence in the current literature. In addition to a modest reduction observed for measures of adiposity such as weight in kilograms, BMI and waist circumference, our review also found associated reductions in intermediate physiological outcomes such as blood pressure and incidence of type 2 diabetes, which reinforces the findings of other systematic reviews87,88,89 documenting reduction in the need for pharmacologic therapy for blood pressure and glycemic control as a potential clinical benefit of modest weight loss.
Limitations
Most evidence was derived from studies that could not be assessed comprehensively for risk of bias. Two-thirds of the pharmacologic studies had pre-randomization run-in periods that involved low-calorie diets with or without placebo, which may have exaggerated the potential benefits of treatment. Potential reporting bias was identified across a number of outcome and comparison-based study groupings. The relatively high attrition rates in many studies leads to further risk of bias. These methodological limitations reduced the strength of evidence, resulting in moderate- and sometimes low-quality ratings, which reduce confidence in the pooled estimates of effect. Results presented for the secondary outcomes should be interpreted with caution because we only included interventions where the focus was on weight loss. Adverse events may be overestimated; data were extracted as reported even when the connection to the intervention was not clear and even if run-in events were included. The search included papers in English or French only and thus may have missed studies in other languages. Most studies were of relatively short duration (≤ 12 mo), and there was a lack of evidence to address the question of whether (and for how long) weight loss is maintained after interventions are completed.
Conclusion
In summary, modest weight reduction confers clinically important benefits and a substantial reduction in the incidence of type 2 diabetes in prediabetic populations, with the potential to improve population health. Future research should include longer term follow-up to observe maintenance of weight loss, to study the effects of repeated weight loss and regain, and to determine if improvements in health outcomes are related to the intervention apart from weight loss.
Supplemental information
For reviewer comments and the original submission of this manuscript, please see www.cmajopen.ca/content/2/4/E306/suppl/DC1
Acknowledgments
Contributors: Leslea Peirson, Donna Ciliska, James Douketis, Donna Fitzpatrick-Lewis and Parminder Raina were involved in conceptualizing the study, data interpretation and writing the manuscript. Leslea Peirson, Donna Ciliska and Donna Fitzpatrick-Lewis were involved in developing the methods, collecting the data and coordinating the project. Muhammad Usman Ali conducted the data analyses and contributed to writing the manuscript. All of the authors revised the manuscript, approved the final version submitted for publication and agreed to act as guarantors of the work.
Acknowledgments
Disclaimer: The views expressed herein are the opinions of the authors and do not necessarily represent the views of the CIHR.
Acknowledgements: Rachel Warren, Meghan Kenny, Eva Tsakonas and Mary Gauld contributed to the relevance and quality assessment, and data-extraction phases. We are grateful to Maureen Rice for the search, Eva Tsakonas for contextual questions (results not reported here) and to Sharon Peck-Reid for database management and formatting of the report. Sarah Connor Gorber and Amanda Shane (Scientific Officers, Public Health Agency of Canada) contributed to the original protocol development and review of drafts of the technical report. Similarly, Paula Brauer, Maria Bacchus, Neil Bell, Elizabeth Shaw and Harminder Singh (members of the Adult Obesity Working Group of the Canadian Task Force on Preventive Health Care) provided comments on the protocol and initial analyses. The Task Force Chair, Cello Tonelli, provided valuable feedback on the manuscript.
Footnotes
-
Competing interests: Donna Ciliska and Parminder Raina have received grants from the Canadian Institutes of Health Research. James Douketis is a current member of the advisory boards for Bayer, Bristol-Myers Squibb, Sanofi and Pfizer. He was a member of the advisory boards for AstraZeneca, Boehringer Ingelheim, Biotie Therapies, Portola Pharmaceuticals and The Medicines Company. He also received speaker fees from AGEN Biomedical, Ortho-Janssen Pharmaceuticals and Boehringer Ingelheim, as well as a grant from Boehringer Ingelheim. No competing interests were declared by the other authors..
-
Funding: Funding was provided by the Canadian Institutes of Health Research (CIHR; www.cihr-irsc.gc.ca). The CIHR played no role in the study design, data analysis, interpretation or decision to submit the paper for publication. Parminder Raina holds a Tier 1 Canada Research Chair in Geroscience and the Raymond and Margaret Labarge Chair in Research and Knowledge Application for Optimal Aging.
References
- © 2014 Canadian Medical Association or its licensors