Abstract
Background: The use of prasugrel or ticagrelor as part of dual antiplatelet therapy with acetylsalicylic acid after acute coronary syndrome (ACS) improves clinical outcomes relative to clopidogrel. The relative cost-effectiveness of these agents are unknown. We conducted an economic analysis evaluating 12 months of treatment with clopidogrel, prasugrel or ticagrelor after ACS.
Methods: We developed a fully probabilistic Markov cohort decision-analytic model using a lifetime horizon, from the perspective of the Ontario Ministry of Health and Long-Term Care. The model incorporated risks of death, recurrent ACS, heart failure, major bleeding and other adverse effects of treatment. Data on probabilities and utilities were obtained from the published literature where available. The primary outcome was quality-adjusted life-years (QALYs).
Results: Treatment with clopidogrel was associated with the lowest effectiveness (7.41 QALYs, 95% confidence interval [CI] 1.05-14.79) and the lowest cost ($39 601, 95% CI $8434-$111 186). Ticagrelor treatment had an effectiveness of 7.50 QALYs (95% CI 1.13-14.84) at a cost of $40 649 (95% CI $9327-$111 881). The incremental cost-effectiveness ratio (ICER) for ticagrelor relative to clopidogrel was $12 205 per QALY gained. Prasugrel had an ICER of $57 630 per QALY gained relative to clopidogrel. Ticagrelor was the preferred option in 90% of simulations at a willingness-to-pay threshold of $50 000 per QALY gained.
Interpretation: Ticagrelor was the most cost-effective agent when used as part of dual antiplatelet therapy after ACS. This conclusion was robust to wide variations in model parameters.
Contemporary guidelines recommend dual antiplatelet therapy with acetylsalicylic acid (ASA) and a P2Y12 receptor antagonist for 1 year after acute coronary syndrome (ACS).1-3 The CURE (Clopidogrel in Unstable Angina to Prevent Recurrent Events) trial showed that clopidogrel reduced adverse cardiovascular events when combined with ASA for 12 months after ACS.1 However, the individual response to clopidogrel is limited by various factors.4 This has prompted research that culminated in the development of prasugrel and ticagrelor, novel P2Y12 receptor antagonists with superior antiplatelet properties compared with clopidogrel.
The TRITON-TIMI 38 trial (Trial to Assess Improvement in Therapeutic Outcomes by Optimizing Platelet Inhibition with Prasugrel-Thrombolysis in Myocardial Infarction 38) showed that use of prasugrel after ACS significantly reduced the risk of recurrent ACS, including stent thrombosis, relative to clopidogrel.5 Similarly, the PLATO (Platelet Inhibition and Patient Outcomes) trial showed that ticagrelor reduced the risk of all-cause death after ACS relative to clopidogrel.6 Both prasugrel and ticagrelor increased bleeding rates, with a more prominent increase in risk with prasugrel.5,6 In addition to these clinical trade-offs, both agents have substantially higher acquisition costs than clopidogrel.7,8
A recent statement from the American College of Cardiology/American Heart Association emphasized the importance of evaluating the clinical benefits of health care interventions in the context of their costs.9 This enables delivery of the highest-quality health care while optimizing scarce resources. Cost-effectiveness analyses have compared clopidogrel individually with prasugrel10 and ticagrelor;11 however, none has directly compared all 3 agents against each other. Decision-analytic modelling is well-suited to addressing this gap in knowledge, because it provides an explicit framework to integrate all available evidence. Accordingly, we conducted an economic analysis comparing the cost-effectiveness of 12 months of treatment with clopidogrel, prasugrel or ticagrelor after an ACS, including ST-segment elevation myocardial infarction (STEMI) and non-STEMI.
Methods
Study design
We developed a fully probabilistic Markov cohort state-transition model, with a life-time horizon. Cycle length was set at 1 month. The model was analyzed from the perspective of the Ontario Ministry of Health and Long-Term Care. The 3 alternatives evaluated in the model were treatment with ticagrelor, prasugrel or clopidogrel for 12 months after revascularization with percutaneous coronary intervention in patients with an ACS.12-14 We expressed effectiveness in terms of quality-adjusted life-years (QALYs) and adjusted costs to 2012 Canadian dollars using the general Consumer Price Index from Statistics Canada.
Incremental cost-effectiveness ratios (ICERs) were calculated by ordering the 3 strategies from lowest to highest lifetime cost, consistent with economic analysis conventions. We determined the ICER based on the incremental cost and effectiveness compared with the next less expensive treatment strategy. If a strategy was more effective than a more expensive alternative, it was a dominant strategy. If the ICER of a strategy was lower than its less expensive alternative, it extendedly dominated that alternative, because it represented more efficient value per unit cost. Based on guidelines, an alternative was considered to be of high value if its ICER was less than $50 000 per QALY gained (1 × per capita gross domestic product [GDP]).9 All utilities and costs were discounted at a rate of 5% per year according to current Canadian recommendations.15
Model structure
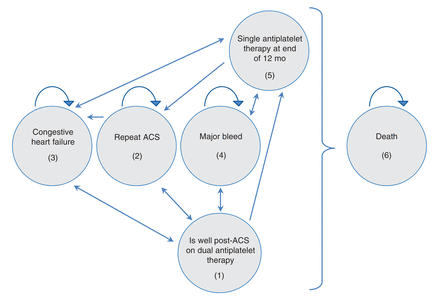
A simplified model schematic is presented in Figure 1. Patients in the model progress through cycles of 1-month duration. All patients begin with dual antiplatelet therapy with ASA, combined with one of clopidogrel, prasugrel or ticagrelor, with the objective of completing 12 months of therapy after ACS. We assumed that every patient had successful revascularization at the time of index percutaneous coronary intervention (PCI) for their ACS.
Simplified schematic of the decision model. This figure illustrates important events and states captured in the model. All patients enter the Markov cohort after percutaneous coronary intervention for myocardial infarction. They receive treatment with acetylsalicylic acid and one of clopidogrel, prasugrel or ticagrelor with the aim of continuing 12 months of dual antiplatelet therapy (1), after which they are transitioned to single antiplatelet therapy (5), if repeat acute coronary syndrome (ACS) does not occur (2). In addition to repeat ACS (2), patients could also have a major bleed (4). Patients could transition to a state of congestive heart failure (3), after which they could persist in that state or recover to being well post-ACS (1). Patients could die (6) at any stage of the model, with a risk dependent on age, antiplatelet therapy exposure, time since last ACS and development of congestive heart failure. Death is by definition an absorbing state.
Within any 1-month cycle, patients could die, develop heart failure or become free of heart failure. Events that occurred within each cycle included recurrent ACS (with possible stent thrombosis), major bleeding or minor adverse effects (minor bleeds, and ticagrelor-associated bradycardia and dyspnea). We factored discontinuation of assigned dual antiplatelet strategy into our model using rates derived from clinical trial data.
We assumed that, if patients had to stop taking clopidogrel within the first 12 months after the index ACS, the drug would be replaced with prasugrel or ticagrelor, with a 50% probability of receiving either agent. If patients were initially prescribed one of prasugrel or ticagrelor and had to stop it within the first 12 months, they were transitioned to clopidogrel. If patients had to stop 2 P2Y12 receptor antagonists, they were classified as intolerant of dual antiplatelet therapy and maintained on single antiplatelet therapy with ASA; such patients were modelled to have higher risks of recurrent ACS and death for the first 12 months after the index ACS.
After 12 months of dual antiplatelet therapy without recurrent events, patients were transitioned to single antiplatelet therapy. If an ACS recurred, patients were transitioned to the last dual antiplatelet strategy they tolerated. Those who were intolerant of dual antiplatelet therapy received angioplasty without stenting and maintained on ASA therapy alone. These patients were modelled to have a higher risk of recurrent ACS for the subsequent 6 months.
Base case
The baseline characteristics of our base case were derived from the weighted means of the characteristics of patients enrolled in the TRITON-TIMI 38, the DISPERSE-2 (the Dose Confirmation Study Assessing Antiplatelet Effects of AZD6140 versus Clopidogrel in Non-ST-segment Elevation Myocardial Infarction) and the PLATO randomized controlled trials.5,6,8,16 The mean age was 62 years (standard deviation 12.5 yr); 61% were male, and 24% had diabetes.
Probabilities
Probabilities were obtained from the published literature and are listed in Table 1.4-6,17-19 All transition probabilities for stent thrombosis, development of heart failure, recurrent ACS and death were time-dependent based on patient age and/or time from the most recent ACS event.
The incidence of recurrent ACS, stent thrombosis and TIMI (Thrombolysis In Myocardial Infarction) major bleeding with clopidogrel was derived from the weighted mean of the event rates in the clopidogrel arm of the trials listed in the base case above. The incidence of these events among patients given prasugrel or ticagrelor was modelled by multiplying the baseline rate in the clopidogrel group with the corresponding hazard ratio (HR) for each event as determined from each agent's phase III trial data. Rates of minor bleeding and other minor adverse effects, as well as rates of discontinuation, were determined directly for each agent with the use the TRITON and PLATO trial data.
The baseline risk of death for patients on ASA monotherapy was derived from age- and sex-specific Ontario life tables and modified by a time-dependent change in the hazard for death based on time since the most recent myocardial infarction20 and the presence or absence of heart failure. In each treatment arm, the risk of death was modified based on the reported HR for death reported in phase III trials of prasugrel, ticagrelor and clopidogrel (Table 2).4-6,20-23
Costs and utilities
Costs and utilities used in our study are summarized in Table 3.24-30 Costs were reported in 2012 Canadian dollars. Unit costs for medications were obtained from the Ontario Drug Benefits Formulary.30 We assumed dispensing fees of once per 3 months (3 months is the maximum time frame that pharmacies prescribe medications under the Ontario Drug Benefit Plan). We used monthly treatment costs of $20.02 for clopidogrel, $80.96 for prasugrel and $90.10 for ticagrelor. We used the Ontario Case Costing Initiative to determine hospital care costs.28 Physician costs were obtained from the 2012 Ontario Schedule of Benefits for Physician Services.29 For each ACS, we assumed there were consultations with an emergency physician, a cardiologist and an interventional cardiologist, a diagnostic angiogram and percutaneous coronary intervention, a transthoracic echocardiogram, and 3 follow-up visits with the attending cardiologist.
We used previously described utility values for the post-MI state, with and without heart failure, as well as for ACS, and major and minor bleeding.15,16,20,24,28-31 The utility of other non-major bleeding-related adverse effects were assumed to be equivalent to those of minor bleeding.22,24,26,28,30-33 The utility of twice-daily dosing, which is necessary for ticagrelor, was assumed to be 0.999.
Statistical analysis
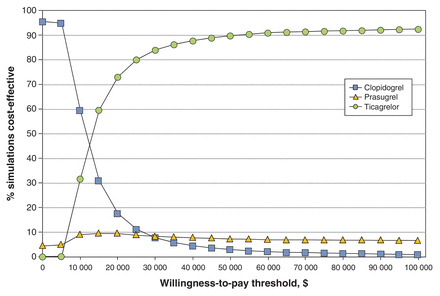
The model was fully probabilistic, with all input parameters (probabilities, utilities and costs) expressed as a distribution, with the mean or expected value and confidence intervals derived from the literature (Tables 1-3). If a confidence interval was not available, we used a conservative estimate of the variance being one-third of the mean.34 We used β distributions for probabilities and utilities, gamma distributions for costs and log-normal distributions for HRs. We ran 10 000 simulations of the model, with parameter values in each simulation based on random draws from each of the distributions in the model. Our final outputs are based on the means of the results from the 10 000 simulations. This probabilistic analysis allows for the full incorporation of the uncertainty of the input parameters. In addition, we expressed the degree of uncertainty by plotting a cost-effectiveness acceptability curve, which illustrates the proportion of the 10 000 simulations in which clopidogrel, prasugrel or ticagrelor was the preferred option at different willingness-to-pay thresholds.
We performed one-way sensitivity analyses on all input parameters, to determine the robustness of our model. The ranges of these sensitivity analyses were based on 95% confidence intervals (CIs) determined by the distributions used for the probabilistic analysis. Given the computation intensity of the probabilistic analyses, one-way sensitivity analyses were conducted deterministically (Appendix 1, available at www.cmajopen.ca/content/3/4/E438/suppl/DC1).
The model was constructed using TreeAge Pro 2013 (TreeAge Software, Inc.). Computation was conducted using 64-core cloud computing with Amazon Web Services.
Results
Base case cost-effectiveness analysis
Clopidogrel treatment resulted in the lowest effectiveness (7.41 QALYs, 95% CI 1.05-14.79) and the lowest cost ($39 601, 95% CI $8434-$111 186). Prasugrel treatment had an effectiveness of 7.43 QALYs (95% CI 1.06-14.79) at a cost of $40 422 (95% CI $9002-$112 574), for an ICER of $57 630 per QALY gained relative to clopidogrel. Ticagrelor treatment had an effectiveness of 7.50 QALYs (95% CI 1.13-14.84) at a cost of $40 649 (95% CI 9327-111 881), for an ICER of $3167 per QALY gained relative to prasugrel. Prasugrel was therefore extendedly dominated by ticagrelor. When compared with clopidogrel, the ICER of ticagrelor was $12 205 per QALY gained (Table 4). Ticagrelor was the preferred agent in 90% of the 10 000 simulations at a willingness-to-pay threshold of $50 000 per QALY. When the threshold was raised to $100 000 per QALY, it was the preferred agent in 92% of the simulations (Figure 2).
Cost-effectiveness acceptability curve from the probabilistic analysis, showing the proportion of 10 000 simulations in which clopidogrel, prasugrel or ticagrelor was the preferred option at different willingness-to-pay thresholds (in 2012 Canadian dollars).
One-way sensitivity analyses
Ticagrelor was the most cost-effective agent throughout the range of most parameters' values in the one-way sensitivity analyses. The conclusion was only sensitive to variations in the value of the HR for death associated with ticagrelor relative to clopidogrel. The ICER associated with ticagrelor relative to clopidogrel exceeded $50 000 per QALY when the HR for death was greater than 0.89. Ticagrelor remained the most cost-effective option when the population was restricted to only younger or older patients.
Interpretation
Our comparison of clopidogrel, prasugrel and ticagrelor suggests that ticagrelor is the most cost-effective P2Y12 receptor antagonist for use in combination with ASA post-PCI after an ACS. This conclusion was relatively robust to variation in the values of important model parameters, with 90% of simulations supporting a preference for ticagrelor.
Clopidogrel is a second-generation thienopyridine that inhibits platelet aggregation. Along with ASA, it has been a mainstay in the management of ACS after PCI. However, a significant proportion of patients who take clopidogrel and ASA after an ACS remain at risk for subsequent adverse cardiovascular events, including death. There are several known drawbacks to clopidogrel. First, it is an inactive prodrug requiring hepatic activation via several cytochrome P450 enzymes.35 This activation takes time, such that maximal platelet inhibition occurs only after 4-5 days.36 Second, the active metabolite irreversibly inhibits the adenosine diphosphate receptor, which may be associated with a greater propensity for bleeding.37 Third, there is substantial individual variability in the platelet inhibitory response to clopidogrel. Prasugrel and ticagrelor are novel P2Y12 receptor antagonists.36,38 Like clopidogrel, prasugrel is a prodrug and an irreversible P2Y12 inhibitor. However, there is less variability in its platelet inhibitory effect.36 Ticagrelor, a reversible and direct-acting oral antagonist of the P2Y12 receptor, provides faster, greater and more consistent P2Y12 inhibition than clopidogrel does.39,40 These pharmacologic properties may help explain some of the different clinical properties of these agents.
Prasugrel substantially reduces the risk of recurrent ACS and stent thrombosis. In fact, an indirect network meta-analysis showed it to be more effective than ticagrelor at reducing stent thrombosis, with an estimated odds ratio of 0.635 (95% CI 0.433-0.932).4 However, this comes at an increased risk of bleeding. The TRITON-TIMI 38 trial indicated that the risk of any bleeding, including TIMI major bleeding, was higher with prasugrel than with clopidogrel, with HRs of 1.46 and 1.31, respectively.5 The aforementioned network meta-analysis suggested that this higher bleeding risk persists in comparisons with ticagrelor, with an odds ratio of 1.43 (95% CI 1.10-1.86).4 Ticagrelor, on the other hand, appears to be less potent than prasugrel with respect to reducing the risk of recurrent ACS and stent thrombosis, but it is associated with a less substantial increase in bleeding risk. Importantly, ticagrelor is associated with a significant reduction in all-cause mortality relative to clopidogrel, with an HR for death of 0.78. This may be a consequence of a more optimal balance between these 2 competing risks. However, other pleiotropic mechanisms, such as promotion of endothelial adenosine activity, have been postulated to explain the survival advantage that is unique to this agent.41
Traditional cardiovascular treatment guidelines have not incorporated resource utilization and value considerations into their recommendations. However, given the finite health care resources available and the increasing costs of providing health services, there has been a recent emphasis on assessment of the cost and value of health care interventions. A recent statement from the American College of Cardiology/American Heart Association emphasized the importance of considering cost and value when making health care decisions in the future of cardiovascular research.9 Although prasugrel and ticagrelor are clearly efficacious, it is currently unclear whether this is offset by their increased cost. The question remains whether they provide added value over clopidogrel.
Although there have been economic analyses assessing clopidogrel, prasugrel and ticagrelor, none has previously compared all 3.10,11,42 It is important to determine the most cost-effective option among these 3 agents, which are all currently available to clinicians and policy-makers. Health care interventions with an ICER of less than $100 000 per QALY are currently defined as providing intermediate value, and those with an ICER of less than $50 000 per QALY are thought to provide high value.9 In our analysis, ticagrelor was the most cost-effective strategy in 92% of the 10 000 simulations at a willingness-to-pay threshold of $100 000 per QALY, and in 90% of the simulations at a threshold of $50 000 per QALY. This means that the model conclusions are stable to a wide range of variability in the parameters used in the model.
Our conclusions support current guidelines for non-STEMI management, which state that it is reasonable to consider ticagrelor in preference to clopidogrel for dual antiplatelet therapy (class IIa indication).43 In contrast, current STEMI guidelines provide no preference for one agent over another.44 Our analysis provides additional cost-effectiveness data to guide decisions by hospitals and third-party payers about the adoption of ticagrelor in lieu of clopidogrel. We anticipate that this information will also be useful to authors of future treatment guidelines.
Limitations
Our study has limitations. First, our model is specific for patients with ACS who undergo PCI. We cannot extrapolate our conclusions to those receiving medical treatment.
Second, our model was a Markov cohort, and therefore it applies to the general population of patients with ACS undergoing revascularization. We did not account for individual patient variability in terms of baseline risk. Such variability may affect the overall cost-effectiveness of an agent. Investigating such subgroups should be a focus for future research.
Third, a potential limitation is that our estimates for efficacy were derived from trials in which either a 300-mg or a 600-mg loading dose of clopidogrel was used. In particular, in the TRITON-TIMI trial that evaluated prasugrel, a 300-mg loading dose of clopidogrel was used in the control arm, whereas a 600-mg loading dose was used in the control arm of the PLATO trial (about 19.6% of the study population) and is used in present-day practice in the setting of primary PCI for STEMI. The difference in death and reinfarction rates between the 2 loading doses of clopidogrel appear to be negligible.45 Moreover, the uncertainty in the efficacy and safety estimates for both ticagrelor and prasugrel has been accounted for in the one-way and probabilistic sensitivity analyses, which showed that our conclusions were robust to wide variations in those numbers.
Fourth, we did not explicitly distinguish between different types of major bleeds in the model. However, we did account for their cumulative impact on mortality and discontinuation rates by using direct estimates of the relative risk of those events from phase III clinical trial data.
Finally, our results and conclusions are based on the current price of the 3 agents in the province of Ontario in 2015. These findings may not be generalizable to other jurisdictions. Furthermore, when the agents come off patent protection, there will likely be a substantial change in price, which would alter our results.
Conclusion
Our economic analysis indicated that ticagrelor is the most cost-effective P2Y12 receptor antagonist when combined with ASA after ACS. These results may aid decision-makers and individual clinicians in both recommending and ultimately selecting the appropriate P2Y12 receptor antagonist in conjunction with ASA for dual antiplatelet therapy after ACS.
Supplemental information
For reviewer comments and the original submission of this manuscript, please see www.cmajopen.ca/content/3/4/E438/suppl/DC1
Footnotes
Competing interests: None declared.
Contributors: Husam Abdel-Qadir and Idan Roifman are joint first authors in this paper. All of the authors contributed to the study concept and design. Husam Abdel-Qadir and Harindra Wijeysundera obtained funding for the study. Harindra Wijeysundera was the study supervisor. All of the authors contributed to the acquisition, analysis and interpretation of data, and the critical revision of the manuscript for important intellectual content. All of the authors approved the final version of the manuscript to be published and agreed to act as guarantors of the work.
Acknowledgements: Husam Abdel-Qadir is supported by a fellowship award from the Canadian Institute of Health Research. Harindra Wijeysundera is supported by a Distinguished Clinician Scientist Award from the Heart and Stroke Foundation of Canada.
Disclaimer: This study was supported by the Institute for Clinical Evaluative Sciences (ICES) which is funded by an annual grant from the Ontario Ministry of Health and Long-Term Care (MOHLTC). The opinions, results and conclusions reported in this article are those of the authors and are independent from the funding sources. No endorsement by ICES ot the Ontario MOHLTC is intended or should be inferred.
References
- Copyright 2015, 8872147 Canada Inc.