Abstract
Background: Hepatitis C virus (HCV) infection causes substantial morbidity and mortality in Canada and is of concern among First Nations communities. In partnership with the Ontario First Nations HIV/AIDS Education Circle, we described trends in HCV testing and epidemiologic features among Status First Nations people in Ontario.
Methods: In this retrospective study, we used health administrative databases for 2006–2014 in Ontario with 3 cohorts of Status First Nations people: those tested for HCV for the first time, those who tested positive for HCV antibodies or RNA, and those with no HCV laboratory or testing records. We examined cohort characteristics, and the annual prevalence and incidence of testing and diagnosis of HCV infection. Outcomes were stratified by region, sex and residence within or outside of First Nations communities.
Results: During the study period, 2423 Status First Nations people were diagnosed with HCV infection, 20 481 received their first test, and 135 185 had no test record. The point prevalence of ever having been tested increased from 6.3 (95% confidence interval [CI] 6.2–6.5) per 100 people in 2006 to 16.2 (95% CI 16.0–16.4) per 100 people in 2014. The point prevalence of diagnosed HCV infection increased from 0.9 (95% CI 0.9–1.0) to 2.0 (95% CI 1.9–2.0) per 100 people. The incidence of first test and of diagnosis increased from 12.1 (95% CI 11.5–12.6) to 21.3 (95% CI 20.5–22.1) per 1000 person-years and from 1.3 (95% CI 1.1–1.5) to 2.3 (95% CI 2.1–2.6) per 1000 person-years, respectively. Testing, diagnosis and prevalence of HCV infection were consistently higher among people living outside of versus within First Nations communities, but larger increases over time were observed among those living within First Nations communities.
Interpretation: Testing and diagnosis of HCV infection increased from 2006 to 2014 among Status First Nations people in Ontario. Our findings indicate the need for population-level efforts to eliminate hepatitis C in First Nations communities.
Hepatitis C virus (HCV) is a leading global cause of morbidity, including chronic hepatic inflammation, cirrhosis, hepatocellular carcinoma and, ultimately, death.1 In 2011, the estimated prevalence of chronic HCV infection in Canada was 0.6%–0.7%, with 44% of cases undiagnosed.2
First Nations people in Canada have a history rooted in colonization and systemic racism that has contributed to health inequities. Hepatitis C has been described as a colonial illness, with risk factors such as injection drug use linked to this history and the trauma caused by colonial stress.3 Higher rates of newly diagnosed HCV infection and younger ages at diagnosis have been observed among First Nations people in Canada, compared to non–First Nations people.3 In 2015, the rate of new HCV cases in 31 First Nations communities in northwestern Ontario was 11 times higher than provincial estimates.4 Such community-specific estimates have limited utility for the diverse First Nations communities throughout Ontario, where more than 200 000 First Nations people reside.5
To fulfill its commitments to the World Health Organization strategy for eliminating viral hepatitis by 2030,6 Canada must prioritize working with Indigenous communities toward HCV infection control.7 The objective of this study was to examine trends in testing and diagnosis of HCV infection among Status First Nations people in Ontario, in partnership with First Nations community organizations.
Methods
Partnership
This study was a participatory research collaboration between First Nations community organizations, ICES and academic researchers. The Ontario First Nations HIV/AIDS Education Circle, an advisory group and partner on this study, met quarterly with the researchers to ensure incorporation of diverse First Nations perspectives and cultural understandings in study conception and design, and data analysis and interpretation. Similar collaborations between First Nations organizations and researchers involving ICES administrative data sets are described elsewhere.8–11
Study design and population
We conducted a population-based retrospective study in 3 cohorts of Status First Nations people aged less than 106 years in Ontario who, from Jan. 1, 2006, to Dec. 31, 2014, received their first HCV test, determined by looking back to 1999 (tested cohort); received at least 1 positive test result (antibody or RNA) for HCV infection (diagnosed cohort);or had no record of an HCV test, determined by looking back to 1999 (no-test cohort). Patients in the tested cohort had to have had their first test in 2006–2014, and the positive test result for patients in the diagnosed cohort could have been after an initial negative result, which could have been before 2006. Therefore, not all people in the diagnosed cohort were nested within the tested cohort.
First Nations people registered under the Indian Act were identified through linkage of the Indian Register to the Registered Persons Database (which contains information on all people registered under the Ontario Health Insurance Plan [OHIP]) at ICES.12 The Indian Register includes demographic and administrative information for all First Nations people who have successfully applied for Indian status under the Indian Act (“Status First Nations”). Access to the Indian Register was granted by the Chiefs of Ontario First Nations Data Governance Committee after the study was reviewed for compliance with the First Nations principles of ownership, control, access and possession (OCAP) and by Grand Council Treaty #3. Permission to access data for members of communities in the Kenora Chiefs Advisory was not granted; as such, they were excluded from the analysis.
We obtained records of HCV testing from Jan. 1, 1999, to Dec. 31, 2014, from Public Health Ontario, a central hub for testing of public health unit specimens. Status First Nations people with HCV testing records were linked to health administrative databases at ICES. In addition to demographic characteristics, data included records of inpatient hospital stays, emergency department visits, same-day surgery and outpatient clinic visits, outpatient diagnostic and laboratory tests, physician services (inpatient, outpatient and long-term care settings), home care, long-term care and complex continuing care for all OHIP recipients, and outpatient drug prescriptions for people aged 65 years or more and other recipients (Appendix 1, available at www.cmajopen.ca/content/9/3/E886/suppl/DC1). These data sets were linked by means of unique encoded identifiers and analyzed at ICES.13,14
To exclude people who left Ontario or died without a death record, we defined a surrogate date of last contact with the health care system as the last date of OHIP eligibility, or 4 years after the last health card use or Public Health Ontario HCV record.
Cohort characteristics
Demographic and clinical characteristics were reported for those who were alive at the reference date. For the diagnosed cohort, the reference date was their laboratory-confirmed diagnosis date. For the tested cohort, it was the date of their earliest sample collection. For the no-test cohort, we used July 1, 2010 (midway through the observation period) as the reference date.
We classified residence according to Ontario health service region (Local Health Integration Network [LHIN]), community size and First Nations community status (within a First Nations community, outside a First Nations community, out of province or unknown) at year of reference, using postal and residence codes from the Registered Persons Database, Discharge Abstract Database and National Ambulatory Care Reporting System (Appendix 2, available at www.cmajopen.ca/content/9/3/E886/suppl/DC1).11
We used International Classification of Diseases, 9th Revision and enhanced Canadian version of the International Statistical Classification of Diseases and Related Health Problems, 10th Revision diagnostic codes from the earliest date available until Dec. 31, 2017, to characterize mental health conditions, HIV infections and hepatitis B virus infections (Appendix 2). We scored multimorbidity 2 years before the reference date using the Johns Hopkins ACG System, Aggregated Diagnosis Groups.15 We identified outpatient prescription drugs for HCV infection from the Ontario Drug Benefit database.
Outcomes
People who had their first record of HCV testing (antibody, RNA, genotyping, viral load) in 2006–2014 were tabulated and characterized as single or repeat testers. We counted people with a positive antibody or RNA test result to calculate the overall proportion of test positivity.
For each year from 2006 to 2014, we counted the number of people who had their first HCV test and the number who received their first positive test result. We used these counts to estimate the annual point prevalence of people ever tested for infection and people diagnosed with infection, by dividing them by the number of Status First Nations people in Ontario alive on Dec. 31 of each year. In addition to date of last contact with the health care system, we used annual postal code to determine Ontario residence on Dec. 31 of each year. In each year, we excluded people whose surrogate date of last contact with the health care system was before Dec. 31 of that year. We used laboratory records from 1999 to 2014 to identify people diagnosed with HCV infection who were still alive for our annual point prevalence estimates. We estimated the annual incidence of first test and of diagnosed infection per 1000 person-years by following each person from Jan. 1, 2006, or date of birth until censoring (date of first test or HCV infection diagnosis, death, date of last contact with the health care system or Dec. 31 of each year).
Statistical analysis
We described continuous variables using mean and standard deviation, or median and interquartile range. We described categoric variables as counts and proportions. Crude point prevalence and incidence measures were standardized by the indirect standardization method.16 For each measure, the internal standard consisted of the 2006 to 2014 combined age-sex distribution of First Nations people tested or diagnosed in comparison to the total First Nations population. Sex-specific measures were age-standardized against the male and female populations. We derived confidence intervals (CIs) using a γ distribution. We conducted all analyses using SAS version 9.4 (SAS Institute) and R version 3.6 (R Core Team).
Ethics approval
This study was approved by the University of Toronto Health Sciences Research Ethics Board.
Results
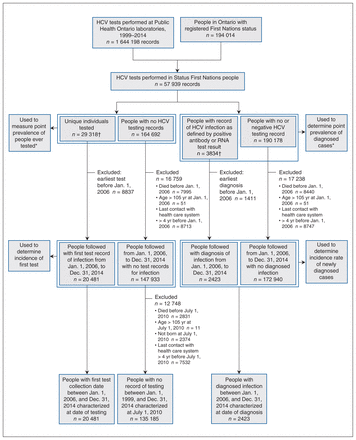
Between 2006 and 2014, 2423 Status First Nations people were diagnosed with HCV infection, 20 481 people received their first HCV test, and 135 185 people had no test record and were alive on July 1, 2010 (Figure 1, Table 1). Of the 2423 people diagnosed with HCV infection, 356 (14.7%) had had a negative antibody test result before diagnosis. Those diagnosed were similar to those tested in mean age (36.2 yr and 34.2 yr, respectively), with most people being younger than 40 years. The majority of people in the diagnosed and no-test cohorts were male (1282 [52.9%] and 69 808 [51.6%], respectively), whereas the majority of those in the tested cohort were female (11 234 [54.8%]). Most people resided outside of First Nations communities.
Flow diagram showing cohort selection. Testing records for the same person on the same day were combined to be a record of a single sample collection. *People who had a testing or diagnosis date in the year of their death still contributed to their respective annual case count for that respective year. †Up to 5 people were excluded because their testing or diagnosis date was more than a year after their death. Note: HCV = hepatitis C virus.
Demographic and clinical characteristics of Status First Nations people in Ontario who were tested for HCV for the first time, tested positive for HCV antibodies or RNA, or had no Public Health Ontario HCV testing records
A total of 201 diagnosed people (8.3%) had a record of HCV-related treatment (Table 1). Also, 188 people (0.9%) in the tested cohort and 35 (0.03%) in the no-test cohort had HCV-related prescriptions.
Higher proportions of diagnosed people than those in the other cohorts had HIV infection, HBV infection and high Aggregated Diagnosis Groups scores (Table 1). Mental health conditions, particularly substance-related and addictive disorders, were common among the diagnosed and tested cohorts (1974 [81.5%] and 10 094 [49.3%], respectively) (Table 1). In the no-test cohort, the most common mental health condition was anxiety disorders (37 263 [27.6%]).
Between their first test and Dec. 31, 2014, 20 467 people received HCV antibody tests (of whom 2073 had a positive result [positivity rate 10.1%]), 1611 received HCV RNA tests (of whom 1030 had a positive result [positivity rate 63.9%]), 993 were tested for viral load, and 963 had genotype testing. A total of 6871/20 467 (33.6%) had repeat antibody testing, and 659/1611 (40.9%) had repeat RNA testing (Appendix 3, Supplemental Table S4, available at www.cmajopen.ca/content/9/3/E886/suppl/DC1).
Incidence and prevalence of testing
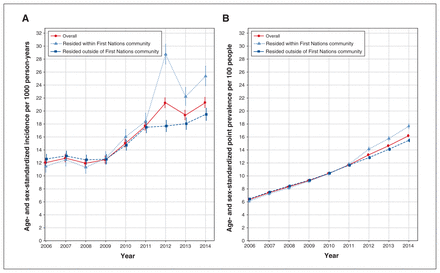
From 2006 to 2014, the annual age- and sex-standardized incidence of first test increased from 12.1 (95% CI 11.5–12.6) to 21.3 (95% CI 20.5–22.1) per 1000 person-years (Figure 2A). Within First Nations communities, it more than doubled, from 11.5 (95% CI 10.6–12.5) to 25.4 (95% CI 23.9–26.9) per 1000 person-years.
(A) Annual age- and sex-standardized incidence per 1000 person-years of first test for hepatitis C virus (HCV) infection among Status First Nations people. (B) Annual age- and sex-standardized point prevalence per 100 people of ever having been tested for HCV infection among Status First Nations people measured at Dec. 31 each year. Error bars represent 95% confidence intervals.
The annual age- and sex-standardized point prevalence of ever having been tested increased from 6.3 (95% CI 6.2–6.5) to 16.2 (95% CI 16.0–16.4) per 100 people (Figure 2B). Within First Nations communities, it increased from 6.1 (95% CI 5.9–6.3) to 17.7 (95% CI 17.3–18.0) per 100 people. Raw counts and annual crude and adjusted measures are reported in Appendix 3, Supplemental Tables S5 to S8. Similar trends were observed across sexes (Appendix 3, Supplemental Figure S1).
Incidence and prevalence of diagnosed infection
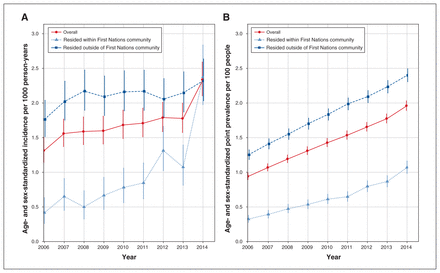
From 2006 to 2014, the annual age- and sex-standardized incidence of diagnosed infection almost doubled, from 1.3 (95% CI 1.1–1.5) to 2.3 (95% CI 2.1–2.6) per 1000 person-years (Figure 3A). Within First Nations communities, it increased nearly sixfold, from 0.4 (95% CI 0.3–0.6) to 2.4 (95% CI 2.0–2.8) per 1000 person-years. By 2014, incidence rates for those within and outside of First Nations communities converged at about 2.3 cases per 1000 person-years. The overall annual age- and sex-standardized point prevalence of diagnosed infection increased from 0.9 (95% CI 0.9–1.0) to 2.0 (95% CI 1.9–2.0) cases per 100 people (Figure 3B). Similar trends were observed across sexes (Appendix 3, Supplemental Figure S2).
(A) Annual age- and sex-standardized incidence per 1000 person-years of diagnosed hepatitis C virus (HCV) infection among Status First Nations people. (B) Annual age- and sex-standardized point prevalence per 100 people of diagnosed HCV infection among Status First Nations people measured at Dec. 31 each year. Error bars represent 95% confidence intervals.
The incidence and prevalence of diagnosed HCV infection were consistently higher for those who resided outside of First Nations communities than for those within First Nations communities (Figure 3), but the differences decreased over time. The incidence was 4 times higher among those who resided outside of versus within First Nations communities in 2006 but almost equal in 2014. The point prevalence was 4 times higher outside of versus within First Nations communities in 2006 but only 2.2 times higher in 2014.
Localization of rates
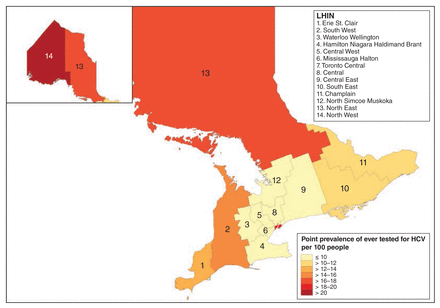
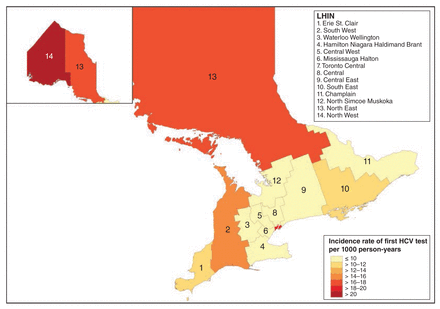
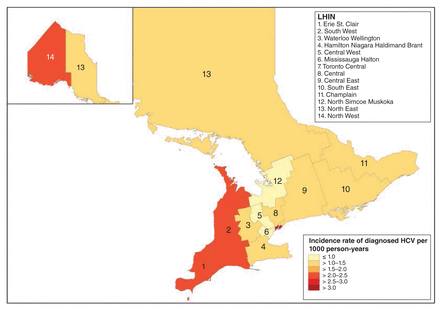
Figures 4 and 5 summarize the age- and sex-standardized point prevalence of ever having been tested and diagnosed HCV infection at Dec. 31, 2014, by LHIN. Both outcomes were higher in the northern LHINs than in the southern LHINs. In the southern LHINs, the prevalence of diagnosed infection was high, particularly in the southwest, whereas the testing prevalence was low. The incidence rates of first test and of diagnosis had localization patterns similar to their point prevalence estimates (Figures 6 and 7).
Age- and sex-standardized point prevalence per 100 people of ever having been tested for hepatitis C virus (HCV) infection among Status First Nations people at Dec. 31, 2014, by Local Health Integration Network (LHIN).
Age- and sex-standardized point prevalence per 100 people of diagnosed hepatitis C virus (HCV) infection among Status First Nations people at Dec. 31, 2014, by Local Health Integration Network (LHIN).
Age- and sex-standardized incidence per 1000 person-years of first hepatitis C virus (HCV) test among Status First Nations people from 2006 to 2014, by Local Health Integration Network (LHIN).
Age- and sex-standardized incidence per 1000 person-years of diagnosed hepatitis C virus (HCV) infection among Status First Nations people from 2006 to 2014, by Local Health Integration Network (LHIN).
Interpretation
This study describes the landscape of HCV testing and infection in Status First Nations people in Ontario from 2006 to 2014. People who were tested for or diagnosed with HCV infection were older, had more comorbidities and mental health issues, and resided outside of rather than within First Nations communities, compared to those never tested. Over the study period, the incidence and point prevalence of both diagnosed infection and testing increased substantially. Larger increases were observed among those who resided within First Nations communities, such that, by 2014, the differences between people living within and outside of communities were smaller.
We found a prevalence of diagnosed HCV infection in 2014 of 2.0% (2 cases per 100 people), whereas the reported national seroprevalence of laboratory-confirmed HCV infection for 2007–2011 was 0.5%.17 The intergenerational impact of colonialism is a critical factor producing this and other health and social inequities in First Nations people.18 The known link between historical trauma and substance abuse — a strong determinant of HCV risk and transmission — is exemplified by the high proportion of people with substance abuse and addictive disorders in our cohorts.19
The increases in point prevalence and incidence of diagnosed HCV infection may be partially explained by higher testing rates. The parallel increases in testing and infection rates in certain LHINs suggest the potential success of partnerships between First Nations communities and their LHINs to formulate accessible, culturally appropriate health care services, especially in mental health and addiction.20 The establishment of programs and initiatives within First Nations communities has increased public awareness of HCV as well as case-finding. Many culturally sensitive harm-reduction initiatives, including counselling and community-based programs to combat opioid dependence and promote community well-being, have been implemented in northwestern Ontario.4,21,22 High rates of testing and diagnosed cases in the North West LHIN provide evidence for the success of such initiatives. Because case-finding has been linked to early treatment and reduction in HCV transmission, increased testing is vital to prevent HCV infection.23
The 2019 Blueprint to Inform Hepatitis C Elimination Efforts in Canada defines how Canada can achieve the targets set by the World Health Organization for hepatitis C elimination.7 First Nations are identified as a priority population for policy and community action around HCV infection.7 Our study aligns with the goals set by the blueprint and provides a comprehensive description of the characteristics and extent of HCV infection in the Status First Nations population in Ontario. With this information, specific interventions can be tailored to the First Nations population to move forward toward hepatitis C elimination.
Limitations
This study reports only part of the picture of the prevalence and incidence of HCV infection diagnosis and testing in the Status First Nations population in Ontario. Many asymptomatic cases remain undiagnosed, and our incidence and prevalence rates of diagnosed infection are almost certainly lower than the true rates. In addition, although standardization of rates aims to reduce bias associated with age and sex distributions of the population, self-selection into the testing group may have introduced another bias.
The Public Health Ontario laboratory data set does not include all results for private diagnostic laboratories, and the Indian Register includes only people who are registered with the Canadian government.11 The Indian Register captured people in ICES administrative data with a linkage accuracy of about 80%.12 This study did not include data for First Nations people who are members of communities in the Kenora Chiefs’ Advisory. First Nations people are eligible for the federal Non-Insured Health Benefits program, which provides prescription drug coverage and federal or band-funded health care services, accessible without an OHIP number. ICES data sets do not capture these services or people without OHIP numbers, so they were not available. Because LHIN and First Nations community residence relied on annual postal and residence codes updated at health card use or renewal, they may have been misclassified.11
The widespread availability of safe and effective short-course direct-acting antiviral HCV treatments in Canada in 2014 has been associated with increases in the number of newly treated people24 and may have increased the incentive for testing. Our HCV testing data ended on Dec. 31, 2014, and did not fully capture these potential consequences.
Conclusion
In partnership with the Ontario First Nations HIV/AIDS Education Circle, and using administrative health data, we identified parallel increases in testing and diagnosis of HCV infection from 2006 to 2014 among Status First Nations people in Ontario. Testing and diagnosis rates were higher outside of than within First Nations communities, but larger increases over time were observed among people who resided within First Nations communities. Canada has prioritized partnerships with First Nations populations to achieve World Health Organization targets for elimination of hepatitis C. Mitigation of infection in these populations requires unique approaches that combine First Nations and Western ways of knowing.
Acknowledgements
The authors thank the Ontario First Nations HIV/AIDS Education Circle (OFNHAEC) and its members for their partnership and insightful comments and thoughtful input throughout this study. They also acknowledge OFNHAEC for their input, permission and guidance in the final dissemination of this project. The authors thank Susan Schultz for developing the methodology to identify First Nations community residence status, and Christina Diong, Ruth Croxford and Graham Mecredy for guidance on the analyses.
Footnotes
Competing interests: William Wong and Murray Krahn report research support from the Canadian Liver Foundation, outside the submitted work. Jordan Feld reports institutional research support from AbbVie, Enanta Pharmaceuticals, Gilead Sciences and Janssen, and consulting fees from AbbVie and Gilead Sciences. No other competing interests were declared.
This article has been peer reviewed.
Contributors: Andrew Mendlowitz and Lyndia Jones conceived the study. Andrew Mendlowitz analyzed the data. Andrew Mendlowitz, Karen Bremner and Murray Krahn drafted the manuscript. All of the authors were involved in designing the study, interpreting the data and revising the manuscript for important intellectual content, approved the final version to be published and agreed to be accountable for all aspects of the work.
Funding: This study was supported by ICES, which is funded by an annual grant from the Ontario Ministry of Health (MOH), and by Canadian Institutes of Health Research Project Grant PJT 166039. This work was also supported by a Canadian Network on Hepatitis C (CanHepC) doctoral trainee fellowship to Andrew Mendlowitz. CanHepC is funded by a joint initiative of the CIHR (NHC-142832) and the Public Health Agency of Canada. This work was also supported by funding from the Canada Research Chairs Program to Murray Krahn, Jennifer Walker and Beate Sander, and an Ontario Early Researcher Award to William Wong.
Data sharing: The data set from this study is held securely in coded form at ICES. Although data-sharing agreements prohibit ICES from making the data set publicly available, access may be granted to those who meet prespecified criteria for confidential access, available at https://www.ices.on.ca/DAS. The full data set creation plan and underlying analytic code are available from the authors on request, with the understanding that the computer programs may rely on coding templates or macros that are unique to ICES and are therefore inaccessible or may require modification.
Disclaimer: The opinions, results and conclusions reported in this article are those of the authors and are independent from the funding sources. No endorsement by ICES or the Ontario Ministry of Health is intended or should be inferred.
Supplemental information: For reviewer comments and the original submission of this manuscript, please see www.cmajopen.ca/content/9/3/E886/suppl/DC1.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/
References
- © 2021 CMA Joule Inc. or its licensors