Abstract
Background: Acceptance of a vaccine against SARS-CoV-2 is critical to achieving high levels of immunization. The objectives of this study were to understand mothers’ SARS-CoV-2 vaccine intentions to explore reasons for and against SARS-CoV-2 vaccination.
Methods: Participants from the All Our Families pregnancy longitudinal cohort whose children had reached ages 9–12 years were invited in May–June 2020 to complete a survey on the impact of COVID-19. The survey covered topics about the impact of the pandemic and included 2 specific questions on mothers’ intentions to vaccinate their child against SARS-CoV-2. Current responses were linked to previously collected data, including infant vaccine uptake. Multinomial regression models were run to estimate associations between demographic factors, past vaccination status and vaccination intention. Qualitative responses regarding factors affecting decision-making were analyzed thematically.
Results: The response rate was 53.8% (1321/2455). A minority of children of participants had partial or no vaccinations at age 2 (n = 200, 15.1%). A total of 60.4% of mothers (n = 798) intended to vaccinate their children with the SARS-CoV-2 vaccine, 8.6% (n = 113) did not intend to vaccinate and 31.0% (n = 410) were unsure. Participants with lower education, lower income and incomplete vaccination history were less likely to intend to vaccinate their children. Thematic analysis of qualitative responses showed 10 themes, including safety and efficacy, long-term effects and a rushed process.
Interpretation: Within a cohort with historically high infant vaccination, a third of mothers remained unsure about vaccinating their children against SARS-CoV-2. Given the many uncertainties about future SARS-CoV-2 vaccines, clear communication regarding safety will be critical to ensuring vaccine uptake.
In Canada, as of May 2021, there have been more than 1 200 000 people infected with SARS-CoV-2 and more than 24 000 deaths due to COVID-19, with more than 3 million deaths worldwide.1,2 Given the human, economic and social cost of the pandemic, the development and uptake of a vaccine remains a critical strategy to mitigate its impact.3 However, the development of a vaccine is not sufficient, as modelling suggests that up to 80% of the population needs to receive a vaccine that is 70% effective to end the pandemic without additional nonpharmaceutical interventions (e.g., physical distancing and masks).4 Vaccine uptake relies on adequate production and distribution, but also on high levels of vaccine acceptance among the general public.5
Emerging studies involving adults suggest that 60%–80% are willing to receive a SARS-CoV-2 vaccine and 10% are not, with the remaining being unsure.5–8 Older age, higher education and higher income are associated with increased willingness to be vaccinated against SARS-CoV-2.5–7 In Canada, the National Advisory Committee on Immunization (NACI) has identified prioritized groups for early vaccination against SARS-CoV-2.9 NACI does not currently identify children as a priority population unless they have other underlying risk factors, as disease burden in children without chronic conditions is low. Currently, one vaccine is approved for children aged 12 and older in Canada (www.canada.ca/en/health-canada/news/2021/05/health-canada-authorizes-use-of-the-pfizer-biontech-covid-19-vaccine-in-children-12-to-15-years-of-age.html); however, it is anticipated that additional vaccines will be approved and younger children will become eligible. If evolving evidence finds that children are important transmitters of infection, vaccination of children will become increasingly important.
Understanding what factors affect decision-making before vaccine rollout in children is critical for early engagement with parents to ensure adequate uptake for SARS-CoV-2 infection control. A limited number of cross-sectional studies, primarily outside of Canada, have asked if parents would be willing to vaccinate their children, with acceptance ranging from 65% to 75%, but motivations for not vaccinating remain understudied.10–12 Past childhood vaccination uptake may be critical to understanding whether parents are willing to accept a new vaccine.
Using longitudinally collected data provides the most accurate description of past vaccination behaviour. Moreover, understanding what factors influence decision-making will be critical to understanding how to communicate about a potential new vaccine. Thus, the objectives of this study were to understand mothers’ SARS-CoV-2 vaccine intentions to explore reasons for and against SARS-CoV-2 vaccination.
Methods
Study design and setting
This study uses data from the All Our Families longitudinal pregnancy cohort study, which began in 2008 with 9 waves of follow-up; details are described elsewhere.13 Participants were originally recruited from the city of Calgary. From May to June 2020, All Our Families participants, whose children had reached ages 9–12 years, were invited to complete a survey on the impact of COVID-19.
Participants
Women were originally eligible if they were less than 25 weeks pregnant, attending prenatal care in Calgary, older than 18 years and spoke sufficient English to complete the survey.13 Participants were originally recruited (2008–2010, baseline) through posters in areas where pregnant women frequent (i.e., low-risk maternity clinics and community centres) and in collaboration with Calgary Laboratory Services, which provides prenatal blood testing for all pregnant women in the city of Calgary.13 Follow-up surveys were conducted twice during pregnancy, at 4 months postpartum, and when the child reached 1, 2, 3, 5 and 8 years of age. Of the 3388 women originally enrolled by telephone, 2455 remain part of the study after 12 years (72.5%) and were invited to participate in the COVID-19 survey.
Data sources
The COVID-19 Impact Survey built on previous waves of data collection and a rapid review of pandemic-related literature and government priorities.14–22 Topic areas were prioritized in consultation with research experts and community groups who had worked with the All Our Families team over the past 12 years. Research experts included pediatricians, psychologists, epidemiologists, members of the COVID-19 Immunity Task Force, nurses and public health experts. Community groups, including Calgary Reads, the First 2000 Days Network and the United Way Council of Champions, provided input on priorities for families during the pandemic.
The survey included both validated measures for mental health (depression and anxiety) and internally developed questions about SARS-CoV-2 infection, job loss, the impact of school closures and physical isolation, and vaccination intention. 23–26 The final set of questions was determined by the core research team and was intended to balance research importance, use of repeated measures from previous data waves, and response burden for mothers. The questionnaire was pilot tested among individuals with children in the target age range for clarity, flow and technical functionality (skip patterns, impossible values), and updated accordingly.
The survey contained about 5 questions per page with a maximum of 27 pages (though shorter for many owing to skip patterns) and took about 30 minutes to complete. The survey was sent to a closed list of eligible participants and administered online using REDCap. Participants were emailed a password-protected unique link to an online survey and entered responses directly. All responses were optional (no forced answers), and participants could review previous answers and could return to the survey as needed until they hit “submit” at the end. Weekly email reminders were sent to participants over 4 weeks, and nonresponders were contacted by telephone. All participants provided informed consent and were provided with a $20 gift card on completion.
The full survey is available in Appendix 1, available at www.cmajopen.ca/content/9/2/E548/suppl/DC1. Data from the survey were stored on a secure server at the University of Calgary and deidentified before analysis. This current study reports the results of the vaccine-intention questions. Specifically, participants were asked, “If an approved COVID-19 vaccine becomes available, would you plan to have your child receive this vaccine?” (no, yes, unsure). Participants had the opportunity to provide a narrative response on what would affect their decision to vaccinate using an open text box. Participant responses were linked to data collected at previous time points. Specifically, demographic information was from the most recent survey (8 yr follow-up survey), except for ethnicity, which was asked at baseline, and income, which was asked on the COVID-19 survey. Infant vaccine uptake was reported by mothers at the 2-year follow-up survey. Maternal report was consistent with administrative vaccination records from public health databases in more than 95% of children.27
Statistical analysis
Quantitative analysis
We provide descriptive statistics on demographic characteristics and responses to the COVID-19 impact survey of the sample. Family income was dichotomized at $80 000, which is when a family of 4 becomes eligible for subsidized rental housing in Calgary.28 To describe which families were least likely to vaccinate, or unsure whether to vaccinate, we estimated a series of multinomial regression models. We examined demographic characteristics (maternal age, income, education, ethnicity and marital status) as well as past vaccination behaviour and possible household exposure to COVID-19. A complete case analysis was used owing to low missing data (< 1%). The reference category was “intend to vaccinate.” All quantitative analyses were carried out using SAS version 9.4.
Qualitative analysis
To understand the factors affecting vaccine intentions, we analyzed the qualitative data from open text responses using a thematic analysis.29 This inductive approach assumes that meaning can be drawn from the content of responses and allows for the generation of greater meaning than content analysis (which counts frequencies of words used).30–32 Following the process outlined by Braun and Clark, 1 author (E.H.) generated the initial codes through data immersion and discussed findings with the team to develop broader themes.29 Codes and themes were refined, and a coding framework was developed with explanatory descriptions. A second author (S.A.E.) reviewed a random sample of 20% of the open text responses and categorized them according to the coding framework.30 The interrater agreement for the categorization of open text responses into themes was 82% (κ 0.76), and differences were resolved through discussion. If participants cited more than 1 reason, their answers were coded to multiple categories.
We used an inductive approach instead of matching responses to previous vaccine hesitancy topics because a SARS-CoV-2 vaccine was hypothetical at the time of data collection. The frequency of responses in themes was counted and categorized by intention to vaccinate. This technique is widely used in larger data sets with short open-ended responses.30–32 The quantification of qualitative data allows for a better understanding of how salient a theme is, and whether it is shared across different vaccine intentions, or if it is only relevant to 1 group.30
Ethics approval
This study received ethical approval from the Conjoint Health Research Ethics Board of the University of Calgary (REB13–0868).
Results
The response rate for the COVID-19 impact survey was 53.8%. Of the 2455 eligible participants, 1321 women responded, and 96.6% provided complete answers. Characteristics of responders and nonresponders can be seen in Appendix 2, available at www.cmajopen.ca/content/9/2/E548/suppl/DC1.
Quantitative results
Participant characteristics stratified by vaccine intention are shown in Table 1. A total of 60.4% (n = 798) of participants reported that they intended to have their 9- to 12-year-old child receive the SARS-CoV-2 vaccine, 8.6% (n = 113) did not intend to vaccinate and 31.0% (n = 410) were unsure. The mean age of mothers was 42 years, 82.1% (n = 1085) had a completed postsecondary degree or higher and 15.0% (n = 198) had household incomes below $80 000.
Characteristics of mothers who participated in a survey on the impact of COVID-19
A total of 15.1% of children of participants had partial or no vaccinations at age 2 (12.0% and 3.2%, respectively). Owing to the low numbers of children with no vaccinations at age 2 (n = 42), vaccine status was categorized as “partial or no vaccination” or “complete vaccination,” according to the infant vaccine schedule in Alberta. Only 1.1% of families had a confirmed SARS-CoV-2 infection at the time of the survey, and another 5.0% had a suspected case.
Unadjusted odds ratios (ORs) describing the associations between participant characteristics and vaccine intentions are presented in Table 2. Lower income was associated with lower intention to vaccinate. Participants with less education were more likely to not want to vaccinate their children (OR 2.80, 95% confidence interval [CI] 1.78–4.40) or be unsure (OR 1.98, 95% CI 1.47–2.71). A similar pattern was seen for income. History of partial or nonvaccination was significantly associated with intent to not vaccinate (OR 2.81, 95% CI 1.78–4.40), but not with uncertainty regarding a SARS-CoV-2 vaccine (OR 1.29, 95% CI 0.92–1.80).
Odd ratios from multinomial models for mothers reporting their intention to have their child receive the SARS-CoV-2 vaccine
Qualitative results
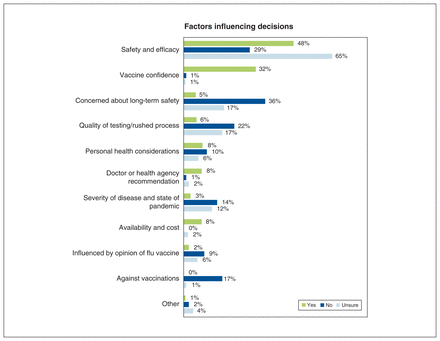
A total of 84.9% of participants (n = 1122) provided a response in the open text box asking about reasons underlying vaccine intention. Using thematic analysis, 10 primary factors influencing decision-making were identified among all mothers, regardless of intention to vaccinate. These themes included safety and efficacy, vaccine confidence, long-term safety, rushed process or scientific quality, perception of personal risk, recommendation from doctor or health authority, perception of risk from COVID-19, availability and cost, attitude toward flu vaccine (both positive and negative), and being against vaccinations. Percentages of respondents in each category (yes, no or unsure) listing a specific factor are presented in Figure 1.
Factors influencing intention to vaccinate among mothers of children aged 9 to 12 years, derived from thematic analysis of open text survey responses from 84.9% of participants (n = 1122).
Example quotes from each theme are provided in Table 3. The most common factor mentioned overall was “safety and efficacy,” which included concerns about potential adverse effects of vaccination. Those intending not to vaccinate specifically noted long-term safety (35.6%) over general safety and efficacy (28.9%). In addition, concerns regarding the rushed nature of testing, which could potentially compromise the safety of the vaccine, was cited among all groups (yes: 5.3%; no: 22.2%; unsure: 16.9%). Personal health conditions were noted among all groups (yes: 8.2%; no: 10.0%; unsure: 6.2%). Overall, 4.0% of participants mentioned their attitude toward the influenza vaccine having an impact on their thoughts on a SARS-CoV-2 vaccine (yes: 2.2%; no: 8.9%; unsure: 6.0%). For example, some who were unsure said they thought the influenza vaccine was ineffective; however, some intending to vaccinate said they got their flu shot every year and would also get a SARS-CoV-2 vaccine.
Quotes for qualitative categories
Interpretation
Among families with generally high levels of complete infant vaccinations, only 60.4% reported that they intended to vaccinate their child against SARS-CoV-2. Findings from both the quantitative and qualitative analysis suggest 3 key messages, outlined in detail below. First, incomplete infant vaccination was associated with negative intentions toward a SARS-CoV-2 vaccine, but not uncertainty. Second, attitudes toward a SARS-CoV-2 vaccine in participants may reflect broader uncertainty about vaccine testing and development. Third, clear communication around SARS-CoV-2 vaccines will be critical to assuage fears about novel vaccines. Understanding parental motivations remains essential for ensuring high uptake if a vaccine is rolled out for this age group.9
Families characterized by less education, lower income and younger age were more likely to have negative or uncertain intentions toward vaccinating their child against SARS-CoV-2, consistent with studies on SARS-CoV-2 vaccine intentions in adults.6–8 A history of partial or incomplete infant vaccination was also associated with not wanting to vaccinate against SARS-CoV-2. Our findings are consistent with those of a multicountry study which found that having their child up to date on childhood vaccines was associated with SARS-CoV-2 vaccine acceptance in parents.12 Our study expands this knowledge by showing that having complete infant vaccinations was not associated with SARS-CoV-2 vaccine uncertainty. Interestingly, 4% of participants mentioned that their thinking about a SARS-CoV-2 vaccine was influenced by their attitude toward the influenza vaccine, with both positive and negative opinions. This suggests that attitudes toward the influenza vaccine may be more salient than attitudes toward childhood vaccines when it comes to SARS-CoV-2 vaccine intentions. The influenza vaccine has historically had lower uptake than childhood vaccines.33 Given the very high proportion of mothers who remain uncertain about a SARS-CoV-2 vaccine, reliance on parental attitudes toward childhood vaccinations may not be sufficient for broad uptake of a SARS-CoV-2 vaccine.
The qualitative data showed that most families had concerns around safety and efficacy of a SARS-CoV-2 vaccine, which has been noted previously.10 However, responses from “no” and “unsure” participants specifically mentioned concerns around long-term safety. Respondents noted the need for years of testing or a guarantee of 100% safety, which may reflect unrealistic expectations for vaccines.34,35 In Canada, vaccine hesitancy has increased in recent years, and careful engagement with those who may be uncertain about vaccines is recommended.36 Moreover, only 1% of “no” participants and 2% of “unsure” participants mentioned willingness to rely on the recommendation from a doctor or public health authority. Longer-term engagement with noncombative strategies involving health care providers and public health leaders may be critical for re-engaging those who remain skeptical about vaccines.36
Finally, among those who did not intend to vaccinate or were unsure, a considerable number cited mistrust or concern with the rushed nature of testing. With the focus on novel vaccine types and approval of 2 messenger RNA (mRNA) vaccines, there is a need to communicate effectively about the development, safety and efficacy of these vaccines.34,37 And although these new developments hold promise, the consequences on overall vaccine confidence could be threatened if novel vaccines have unintended consequences.38,39 Owing to the scope of the pandemic, vaccine trials are increasingly being highlighted in mainstream media, and data from Canada and Australia suggest that acceptance of a new SARS-CoV-2 vaccine continues to change.40–42 Clear communication around risks and benefits is critical, and research into effective communication strategies around novel vaccines is urgently needed.34,43
Limitations
This study has some limitations. This survey had a response rate of 54%, which is lower than response rates for other waves of data collection in this cohort, but is high for participants facing a crisis situation.13,44,45 Other research in this area generally uses self-selection techniques, which can result in unrepresentative samples wherein the direction of bias is difficult to estimate.46 In our study, respondents were generally more affluent and more educated than nonrespondents, and had higher complete infant vaccination (85%) than the average in Alberta (71%).47 We expect that this would underestimate attitudes against a SARS-CoV-2 vaccine and could bias associations toward the null, meaning our findings are generally conservative.
Our analysis approach provides overall estimates that may not fully capture subgroup differences related to income and vulnerability. The number of participants who did not intend to vaccinate was 113 (9%), which resulted in reduced precision. Our data do not include paternal opinions, as fathers were not enrolled in the original All Our Families study. We examined intentions across several participant characteristics, but did not have comprehensive information on child chronic conditions, which could affect vaccine intention. Qualitative responses are preliminary, and more in-depth qualitative research is needed. Data were collected during the first wave of COVID-19 (May–June 2020). As the length and severity of the COVID-19 pandemic increases and vaccine trials and approvals evolve, early vaccine intentions may change over time. We had validated information for vaccination status only up to age 2 and no information on flu vaccine uptake.
Conclusion
In this sample, 60.4% of mothers surveyed in May and June 2020 indicated that they were willing to vaccinate their child against SARS-CoV-2 should a vaccine be approved for children; 31.0% said they were unsure and 8.6% said they were unwilling. Families with lower income or lower education may be more reluctant to accept a SARS-CoV-2 vaccine. Complete infant vaccination did not differ between mothers who intended to vaccinate against SARS-CoV-2 and mothers who were unsure, suggesting that mothers may view a novel SARS-CoV-2 vaccine differently from traditional infant vaccinations. Moreover, our population had higher average rates of complete infant vaccination than the average in Alberta, suggesting that positive SARS-CoV-2 vaccination intentions may be even lower in Alberta than our reported 60.4%. To maximize children’s uptake of a SARS-CoV-2 vaccine, assuaging concerns regarding safety, efficacy and testing appears to be paramount. Targeted public health strategies that include clear communication about safety and efficacy may increase acceptance. Emphasis on quality of scientific evidence may be particularly salient among mothers who are unsure.
Acknowledgments
The authors acknowledge the All Our Families research team and thank the participants who took part in the study.
Footnotes
Competing interests: Suzanne Tough, Sheila McDonald, Erin Hetherington and Sarah Edwards report grants from the Canadian Institutes of Health Research (CIHR) and from Alberta Innovates during the conduct of the study.
This article has been peer reviewed.
Contributors: Erin Hetherington and Sarah Edwards conceptualized and designed the study and data collection instruments, acquired funding, analyzed the data and wrote the initial manuscript. Sheila McDonald and Suzanne Tough conceptualized and designed the study and acquired funding, coordinated and supervised data collection on this study and the historical cohort study, and reviewed and revised the manuscript. Nicole Racine, Shannon MacDonald and Sheri Madigan assisted in the interpretation of results and critically reviewed and revised the manuscript. All authors gave final approval of the version to be published and agree to be accountable for all aspects of the work. Sheila McDonald and Suzanne Tough contributed equally as co–senior authors.
Funding: Funding for this project was provided by the Canadian Institutes of Health Research Canadian 2019 Novel Coronavirus (COVID-19) Rapid Research Funding Opportunity and Alberta Innovates. The All Our Families cohort is funded through Alberta Innovates Interdisciplinary Team Grant #200700595 and the Alberta Children’s Hospital Foundation. The funders and sponsors did not participate in the work.
Data sharing: Individual, deidentified data are available to researchers who provide a methodologically sound proposal for research. Inquiries regarding access to data dictionaries, study protocols and questionnaires may be made at https://allourfamiliesstudy.com/data-access.
Supplemental information: For reviewer comments and the original submission of this manuscript, please see www.cmajopen.ca/content/9/2/E548/suppl/DC1.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/
References
- © 2021 CMA Joule Inc. or its licensors