Abstract
Background: A massive hemorrhage protocol (MHP) enables rapid delivery of blood components in a patient who is exsanguinating pending definitive hemorrhage control, but there is variability in MHP implementation rates, content and compliance owing to challenges presented by infrequent activation, variable team performance and patient acuity. The goal of this project was to identify the key evidence-based principles and quality indicators required to develop a standardized regional MHP.
Methods: A modified Delphi consensus technique was performed in the spring and summer of 2018. Panellists used survey links to independently review and rate (on a 7-point Likert scale) 43 statements and 8 quality indicators drafted by a steering committee composed of transfusion medicine specialists and technologists, and trauma physicians. External stakeholder input from all hospitals in Ontario was sought.
Results: Three rounds were held with 36 experts from diverse clinical backgrounds. Consensus was reached for 42 statements and 8 quality indicators. Additional modifications from external stakeholders were incorporated to form the foundation for the proposed MHP.
Interpretation: This MHP template will provide the basis for the design of an MHP toolkit, including specific recommendations for pediatric and obstetrical patients, and for hospitals with limited availability of blood components or means to achieve definitive hemorrhage control. We believe that harmonization of MHPs in our region will simplify training, increase uptake of evidence-based interventions, enhance communication, improve patient comfort and safety, and, ultimately, improve patient outcomes.
Massive bleeding is a leading preventable cause of death following trauma, childbirth and surgery.1–3 There were 5.1 million deaths after traumatic injury worldwide in 2010, mostly affecting young people, accounting for nearly 10% of all deaths.4 In the United States, it is estimated that up to 20% of such deaths are the direct result of preventable hemorrhage.4–7 Management of unstable hemorrhagic shock is centred on stabilizing the patient with prompt transfusion of blood components, and rapid identification and treatment of the source of bleeding. Patient outcome is dependent on the availability of rapid definitive surgical intervention, support of a transfusion medicine and clinical laboratory, prompt access to hemostatic agents and care provided by a high-performing interdisciplinary team.8 In the trauma literature, protocolized delivery of massive transfusion streamlines the complexities of rapid access to surgical care and blood components, decreases variability of treatment, increases protocol compliance, reduces blood component wastage, facilitates interprofessional communication and allows for tracking of metrics for continuous quality improvement.9–12
Most academic institutions have a massive hemorrhage protocol (MHP) in place to rapidly deliver blood components and coordinate care in the setting of traumatic injury.13 However, in a recent survey of 150 hospitals in Ontario, the proportion of hospitals with MHPs in nonacademic settings was lower than that in academic settings, and there was significant protocol heterogeneity between hospitals.14 This was also seen in well-established trauma centres in the US,15 where civilian implementation is still highly variable.12 Compliance with MHPs is also highly variable,12 and low compliance may have ramifications for patient outcomes.16 A systematic review of before–after studies comparing patients with trauma managed in periods with and without an MHP showed an association between MHP use and better survival. 17 The benefits of an MHP have not been tested in rigorous prospective randomized trials. Maintaining high levels of compliance with MHPs appears to be a universal challenge. 12,16 Ontario has the largest population of all the Canadian provinces and provides hospital care in a diverse mixture of settings with high and low health care resources. Massive hemorrhage is an infrequent event in many areas served by small regional hospitals. As a result, access to blood components, laboratory tests of hemostasis and surgical expertise are highly variable. Instituting an adaptable MHP for the province based on the local health care resource setting is needed to streamline the complex logistics of rapid delivery of blood components, facilitate rapid patient transfer where required and reduce the cognitive burden on bedside clinicians. With the ultimate goal of developing such a protocol, we aimed to identify the key principles and quality indicators required to develop a provincial standardized evidence-based MHP template for hospitals.
Methods
Steering committee and panellists
A steering committee composed of transfusion medicine specialists and technologists, and trauma physicians selected the panel members, organized the MHP forum, administered the surveys and analyzed the results. The steering committee assembled a panel of 36 content experts to represent relevant stakeholders. The panel members were selected through the Ontario Regional Blood Coordinating Network, which oversees transfusion use, audits of practice and educational initiatives for the region. The steering committee selected members for their broad expertise and responsibility in administering MHPs across Ontario’s geographically diverse network. Panel participation was voluntary and not financially remunerated. Travel reimbursement and meals were provided, and authorship was afforded only to those who participated in all phases of the Delphi process. The panel included anesthesiologists, trauma surgeons, obstetricians, hematologists, transfusion medicine physicians, emergency physicians, prehospital and transport medicine physicians, intensivists, blood supplier representatives, nurses, technologists and a patient. The group represented the geographically diverse health care programs in the province, with representatives from academic hospitals, specialized pediatric institutions, suburban hospitals and smaller, rural hospitals.
The panellists were informed of the purpose and scope of the exercise, and of the requirement to attend a 2-day MHP forum in Toronto and complete all rounds of the Delphi exercise. To standardize the knowledge base of panellists, they were provided with copies of original papers selected by the steering committee and reflecting the most up-to-date evidence in the area of massive hemorrhage management before the first round. The MHP forum had both didactic and interactive sessions, in which content experts reviewed each area of the MHP for 15 minutes, after which there was a 15-minute discussion period for the attendees.
Study design
We chose a modified Delphi technique to establish consensus and the framework for the provincial MHP toolkit. The Delphi technique is a systematic, interactive method that relies on a panel of experts to converge on consensus statements following a series of iterative surveys.18 Rounds of surveys are continued until consensus is achieved. The Delphi technique is deemed a relevant source of evidence in health care research and is particularly important if randomized controlled trials are unavailable to set health care policies.19 Our group used this method previously20 and noted its ability to cost-effectively include a large number of participants who are geographically dispersed, while preventing unequal representation or biasing of opinions in the consensus through iterations of anonymous voting.21 We modified the Delphi technique to allow for open forum of discussion in round 1 only, to ensure that the broadest range of expert experience was captured in the consensus.
The modified Delphi rounds were conducted independently by each panellist via an emailed survey link (Lime-Survey). Survey responses were anonymized before centralized review. Each statement was independently rated on a 7-point Likert scale from “Definitely should not include” to “Definitely should include” in the MHP, with cut-offs based on previously established Delphi criteria.20 There was an “opt out” option for each statement to account for possible lack of expertise in a specific area (“Unable to rate as outside area of expertise”). Panellists were asked to provide suggestions to enhance statement clarity with each numerical ranking. Panellists were instructed to answer questions on the basis of what they considered optimal patient care or best practices, rather than what they believed was currently operationally feasible at their institution.
The first round consisted of 43 statements and 8 quality indicators that had been drafted by the steering committee based on available literature and existing MHPs. The statements were based on the core tenets of existing MHPs, such as rapid and reliable access to blood components, tertiary care transfer and use of antifibrinolytic agents, and the quality indicators were based on quality measures established by the National Surgical Quality Improvement Program. The first round was conducted 1 month before the MHP forum. The survey responses of the first round were anonymized and presented to the panellists and the steering committee on day 2 of the MHP forum for discussion of the phrasing and structure of statements scoring below a median Likert score of 5.5 (see a priori criteria below), as the modification to the Delphi methodology. Round 1 provided panellists with the ability to add statements and quality indicators. To ensure nonbiased and independent statement review, panellists were not provided the ratings and survey comments of the other panel members. After round 1, no further inperson meetings or formal interactions between panellists took place.
Criteria for disposition of items
A priori criteria for disposition of the items in the first round were established as follows:
Items receiving a median Likert score of at least 5.5 (out of 7) would be accepted as written and not subject to further rounds. These statements were to be incorporated into a provincial MHP as written, unless a clear improvement in phrasing was suggested by a panellist that would not change the intent of the item.
Items with a median score of 2.6 to 5.4 were to be discussed at the in-person meeting with all the panellists. Following discussion, the items would be revised by the steering committee and sent out electronically in the second round.
Items with a median score of 2.5 or less were to be removed from further rounds, unless there was strong opposition by the panel, in which case a revision would be drafted for the second round.
Panellists were provided with a comment box to allow for addition of novel statements and quality indicators on the first round and were allowed to suggest additional statements and quality indicators at the in-person meeting. No additional statements were added after round 2.
A priori criteria for disposition of the items in the second or later round were established as follows:
Items receiving a median Likert score of at least 5.5 would be accepted as written and not subject to further rounds, unless a clear improvement in phrasing was suggested by a panellist that would not change the intent of the item.
Items with a median score of 2.5 to 5.4 were to be rewritten on the basis of comments by the panellists and sent out in the third or subsequent round.
Items with a median score of 2.4 or less were to be removed from further rounds of scoring.
Where suggested by panellists and/or the steering committee, merging or division of statements could occur where appropriate.
After consensus was reached in the final Delphi round, the statements were circulated via email to the medical directors of transfusion medicine responsible for 150 of the 262 Ontario hospitals with licensed transfusion laboratories. The accompanying letter included a request to distribute the statements to members of their hospital transfusion committees and MHP leaders for feedback. These 304 people represented the clinical and technical leads of the laboratories. All hospitals in Ontario able to issue red blood cells have a medical director who is registered with the provincial Ministry of Health and Long-Term Care, which provided us with an up-to-date email contact list. Feedback was collated and recommendations were incorporated by the steering committee where necessary to improve the clarity of the statements and their justification.
The initial drafting of the consensus statements was completed by March 2018. Round 1 of the consensus panel was completed on Apr. 13, 2018, and was discussed in person on Apr. 21, 2018 in the MHP forum. Rounds 2 and 3 were subsequently completed in June 2018 and August 2018, respectively.
Ethics approval
Ethics approval was not required for this Delphi exercise.
Results
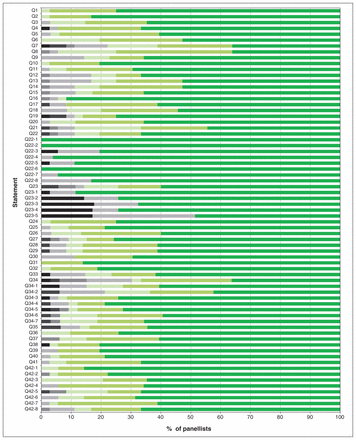
Of the 44 experts invited to participate, 36 agreed (Appendix 1, available at www.cmajopen.ca/content/7/3/E546/suppl/DC1). A 100% response rate was achieved from panellists in all 3 rounds of the modified Delphi exercise. The progression toward consensus based on Likert scores is shown in Figure 1, and the overall degree of consensus for each statement by the last round is shown in Figure 2. Consensus was achieved for all statements by round 3, yielding a final consensus document with 42 statements and 8 quality indicators. Owing to lack of consensus on round 1, statements 7, 22, 23 and 34 were broken into their individual components for rounds 2 and 3 to assist with understanding the components driving the lack of consensus (Figure 1). Unless otherwise specified, all statements and quality metrics also apply to a pediatric MHP. The statements together with a brief rationale for each are presented in Table 1, in a logical order rather than in order of clinical importance.
Median scores on a 7-point Likert scale for the 3 rounds of the Delphi exercise. Counts are provided for cases in which panellists were asked to rank or choose between options rather than use the Likert scale. Green = passed with minor or no phrasing adjustment (empty green box = passed on a preceding round); red = statements that did not pass based on numerical scoring and/or critical written comments, resulting in a major content revision; gray = statements that were not accepted and were then merged for the subsequent round; blue = a new statement that was added after round 1. Split rows denote the need for division of the statement into its components for the scoring round. Note: CBC = complete blood count, iCalcium = ionized calcium, INR = international normalized ratio, NA = not applicable, aPTT = activated partial thromboplastin time.
Proportion of Likert scores at the time of the final consensus round for each statement. Panellists were asked to indicate agreement on a 7-point Likert scale (7 = highest level of agreement). Black and gray = scores less than 5; green shades = scores of 5 or more. Responses of “no,” “uncertain” and “yes” are denoted as black, gray and green, respectively.
Massive hemorrhage protocol statements and rationale
Quality metrics to be tracked on all activations of the protocol
| Quality metric | Local reporting | Provincial reporting |
|---|---|---|
| Q1. Proportion of patients receiving tranexamic acid within 1 h of protocol activation | X | X |
| Q2. Proportion of patients in whom RBC transfusion is initiated within 15 min of protocol activation | X | X |
| Q3. Proportion of patients (of patients requiring transfer for definitive care) with initiation of call for transfer within 60 min of protocol activation | X | |
| Q4. Proportion of patients achieving temperature ≥ 35°C at termination of the protocol | X | |
| Q5. Proportion of patients with hemoglobin levels maintained between 60 and 110 g/L during protocol activation, excluding certain pediatric populations (e.g., neonates) that may require higher hemoglobin values | X | |
| Q6. Proportion of patients transitioned to group-specific red blood cells and plasma within 90 min of arrival/onset of hemorrhage | X | X |
| Q7. Proportion of patients with appropriate activation (≥ 6 RBC units in first 24 h, > 40 mL/kg per 24 h of RBCs in pediatric patients) or before this level in patients dying due to hemorrhage within 24 h | X | |
| Q8. Proportion of patients without any blood component wastage (including plasma that is thawed and not used within the 5-day limit on another patient) | X |
Four areas required additional rounds and major modifications: 1) selection of the name of the protocol, 2) selection of the laboratory resuscitation targets, 3) determination of the pack configurations and 4) clarification of the role of recombinant factor VIIa. The primary obstacle to selecting a name for the protocol was that many hospitals already had long-standing MHPs with specific names. Consensus on the laboratory targets and pack configuration was achieved in the third round by splitting statements into subsections. The recombinant factor VIIa statement required 3 rounds of review to ensure that the phrasing mitigated the panellists’ apprehensions regarding this controversial therapy. Following the generation of consensus statements, no substantive changes to the statements were made, but modifications were required to clarify the supporting text.
The external review by the medical directors of transfusion did not necessitate changes to the statements; however, the feedback provided valuable input to strengthen the supporting text and provide input on logistical and implementation challenges. All statements and the contents of the manuscript were approved by all authors and panellists.
Interpretation
Through a modified Delphi iterative process, we selected and refined 42 statements and 8 quality indicators to form the foundation for the proposed provincial standardized, evidence-based MHP template for hospitals. The MHP toolkit will include training material, simulation exercises, checklists, template policies and procedures, and patient material. In addition to decreasing variability in care, reducing cognitive load on providers, improving communication between the clinical and laboratory teams, increasing uptake of evidence-based treatments and ultimately improving patient outcomes, we hope that this initiative will improve patient comfort and safety, communication with families and disclosure of transfusion risks to patients. Although the lists of statements are not exhaustive, we hope that they address the current widespread variability in MHP structure.14,15 Both the modified Delphi process and the external stakeholder consultation assisted with statement construction to ensure clarity for both experts in transfusion medicine and health care personnel working outside of the laboratory.
This consensus document created by the modified Delphi method is a powerful tool that is broadly applicable and adaptable to many hospital settings. The consensus panel included excellent representation by resource-limited and geographically constrained sites in rural Ontario, as well as resource-rich tertiary care centres. As a result, the recommendations reflect best practice but are also flexible, as they include modifiers for clinical environments with fewer team members and limited access to laboratory testing and blood product availability. For example, the MHP recommends that centres without the ability for definitive hemorrhage control call for early transport (statements 1 and 10), facilities where certain blood components are limited are offered alternatives (statement 34), and there is dosage modification for the initial antifibrinolytic agent for patients in remote locations to ensure administration of a timely and complete dose (statement 40). In comparison, a recent survey of hospitals with MHPs in the United States showed that the majority were in large academic centres, calling for a higher ratio of plasma:red blood cells (1:1), with very few unified MHPs in place to encompass nontrauma indications.101
The Delphi technique was chosen as the method to develop consensus statements from expert stakeholders because of the absence of clinical trial evidence for all aspects of the management of the massively bleeding patient.102 Although clinical trials provide considerable guidance on the utility of blood component ratios,80 antifibrinolytic agents,93,94 use of recombinant factor VIIa84,85 and other areas of management, they fail to provide recommendations on how to construct the protocol, modifications for community hospitals or specific patient populations.9,12,26 We modified the Delphi exercise to allow for an open forum after round 1 to increase input from experienced practitioners and to discuss the vast quantity of available literature to ensure that all participants had a foundation in MHP knowledge. This allowed for the broadest capture of areas of massive hemorrhage that are logistical in nature and do not lend themselves to evaluation in clinical trials, such as communication strategies and procedures, protocol nomenclature, frequency and type of laboratory testing, laboratory resuscitation targets, and blood component transport and bedside storage.26 Importantly, modification of the Delphi exercise also allowed the invaluable input of a patient representative, who provided insight into the importance of patient-related outcomes such as communication and hypothermia management to ensure comfort.
Limitations
The major limitations and challenges of our modified Delphi process included the limited number of panellists per specialty and hospital type (e.g., obstetrics, remote hospitals) owing to cost and logistical limitations. Furthermore, 1 panellist ranked the statements based on feasibility and/or cost of recommendations despite instructions to rank based on best practices; however, later in round 1, the panellist reranked based on best practices. Another limitation was the failure to include panellists involved in hospital blood/sample transportation and communications, as these colleagues are critically important to the success of the MHP (e.g., ensuring a constant supply of blood packs from the bank). Given the numerous items lacking clinical trial evidence, our recommendations will need to be revisited and updated at regular intervals to evolve with this rapidly changing field of medicine. We note a particular lack of high-quality evidence for pediatric patients owing to exclusion of these patients from many clinical trials.
Conclusion
We are confident that the 42 statements and 8 quality indicators, constructed through a highly structured process and with the involvement of a diverse and knowledgeable group of experts, will serve as a strong foundation for the creation of a robust MHP toolkit. The MHP toolkit, currently under development, will consist of recommendations and educational resources for 12 sections (patient transport; principals of damage control resuscitation; team dynamics; communication; laboratory testing; temperature management; use of blood components and adjuncts, including anticoagulant reversal; simulation and training tools; pediatric-specific recommendations; patient communication follow-up tools; research goals; and quality metrics and reporting) written by an expert working group, to be published and freely accessible in the spring of 2020 on the Ontario Regional Blood Coordinating Network website (http://transfusionontario.org/en/) and presented at the network’s spring Transfusion Committee Forum. We expect that, with the use of the toolkit, hospitals will achieve higher adoption of evidence-based care of the patient with massive hemorrhage, improved speed of delivery of blood components and hemostatic adjuncts, and more diligent monitoring of clinical and laboratory parameters. There is an opportunity to track patient outcomes in existing prospectively collected databases in trauma and obstetrics and for inpatients held by the Canadian Institutes for Health Information in Ontario to understand the impact of this effort to standardize the care of these complex, high-acuity patients.
Footnotes
Competing interests: For panellists and conflicts of interest, see Appendix 1 (available at www.cmajopen.ca/content/7/3/E546/suppl/DC1).
This article has been peer reviewed.
Contributors: All of the steering committee members contributed to the study design and data analysis and interpretation, drafted the manuscript and critically revised it for important intellectual content. All of the panel members contributed to data interpretation, drafted the manuscript and critically revised it for important intellectual content. All of the authors approved the final version to be published and agreed to act as guarantors of the work.
Funding: This work was supported by the Ontario Regional Blood Coordinating Network through the Ontario Ministry of Health and Long-Term Care and Canadian Blood Services.
Supplemental information: For reviewer comments and the original submission of this manuscript, please see www.cmajopen.ca/content/7/3/E546/suppl/DC1.
References
- Copyright 2019, Joule Inc. or its licensors