Abstract
Background: With combination antiretroviral therapy (ART) and increased longevity, cancer is a leading cause of morbidity among people with HIV. We characterized trends in cancer burden among people with HIV in Ontario, Canada, between 1997 and 2020.
Methods: We conducted a population-based, retrospective cohort study of adults with HIV using linked administrative health databases from Jan. 1, 1997, to Nov. 1, 2020. We grouped cancers as infection-related AIDS-defining cancers (ADCs), infection-related non-ADCs (NADCs) and infection-unrelated cancers. We calculated age-standardized incidence rates per 100 000 person-years with 95% confidence intervals (CIs) using direct standardization, stratified by calendar period and sex. We also calculated limited-duration prevalence.
Results: Among 19 403 adults living with HIV (79% males), 1275 incident cancers were diagnosed. From 1997–2000 to 2016– 2020, we saw a decrease in the incidence of all cancers (1113.9 [95% CI 657.7–1765.6] to 683.5 [95% CI 613.4–759.4] per 100 000 person-years), ADCs (403.1 [95% CI 194.2–739.0] to 103.8 [95% CI 79.2–133.6] per 100 000 person-years) and infection-related NADCs (196.6 [95% CI 37.9–591.9] to 121.9 [95% CI 94.3–154.9] per 100 000 person-years). The incidence of infection-unrelated cancers was stable at 451.0 per 100 000 person-years (95% CI 410.3–494.7). The incidence of cancer among females increased over time but was similar to that of males in 2016–2020.
Interpretation: Over a 24-year period, the incidence of cancer decreased overall, largely driven by a considerable decrease in the incidence of ADC, whereas the incidence of infection-unrelated cancer remained unchanged and contributed to the greatest burden of cancer. These findings could reflect combination ART–mediated changes in infectious comorbidity and increased life expectancy; targeted cancer screening and prevention strategies are needed.
Cancer is an important comorbidity among people living with HIV, given increased longevity owing to combination antiretroviral therapy (ART).1–4 The risk of cancer is reportedly higher in people with HIV than the general population,5,6 reflecting a complex interplay between various component causes of cancer, including HIV-related immunosuppression,7 decreased immune surveillance of oncogenic coinfections8 and a higher prevalence of known cancer risk factors such as smoking and alcohol use.9 In large studies in North America and Europe, rates of AIDS-defining cancers (ADCs) — including Kaposi sarcoma, non-Hodgkin lymphoma and invasive cervical cancer — declined markedly in the years after the introduction of combination ART. Non-ADCs (NADCs) now constitute most cancers among people with HIV, with an estimated 2-times higher risk of NADC6,10 in the ART era compared with pre-ART.3,6,11,12 Specifically, rates of infection-related NADCs have increased, including anal cancer related to human papillomavirus (HPV), and liver cancer related to hepatitis B and C.5,6 Similarly, in British Columbia from 1997 to 2018, rates of ADCs declined from 15.4 to 3.0 per 1000 person-years and infection-unrelated cancers rose from 1.2 to 4.1 per 1000 person-years.13 In Ontario, cancer was the most common cause of death among people with HIV after HIV/AIDS from 1995 to 2014.4
As the epidemiology of cancer among people with HIV evolves, it is crucial to produce timely population-based estimates to inform prevention and care service planning.5,6 However, most North American studies quantifying cancer trends among people with HIV have been conducted in the United States,5,6,10–12,14–19 with few in Canada.13,20,21 Findings from the US may not be generalizable to Canada, where cancer diagnosis and treatment is less likely to be influenced by disparities in health insurance status, given its publicly funded health insurance program for medically necessary services22,23 and screening programs for breast, cervical and colorectal cancers in most provinces and territories.24,25 Equally important is to have local, context-specific data to guide medical care and shared clinical decision-making at the community level. Historical differences in HIV epidemics and modes of HIV acquisition may influence cancer patterns based on the local epidemiology of coinfection with oncogenic infectious agents. For example, in BC, a higher proportion of people with HIV acquired HIV infection through sharing of injection drug equipment, suggesting higher risk for cancers caused by hepatitis viruses, whereas in Ontario, where sexual transmission is a more common mode of HIV acquisition, HPV-related cancers may be more likely.26
Ontario is the province with the highest number of people living with HIV in Canada; in 2017–2018, over 40% of new HIV infections in Canada occurred in Ontario.27,28 Therefore, we sought to estimate the incidence and prevalence of cancer from 1997 to 2020 among people with HIV in Ontario.
Methods
Study design and setting
We conducted a retrospective cohort study of cancer trends, from Jan. 1, 1997, to Nov. 1, 2020, among people with HIV using linked administrative health data sets available at ICES, an independent, nonprofit research institute whose legal status under Ontario’s health information privacy law allows it to collect and analyze health care and demographic data, without consent, for health system evaluation and improvement. The study was reported using the Reporting of Studies Conducted Using Observational Routinely-Collected Health Data (RECORD) checklist.29
Participants
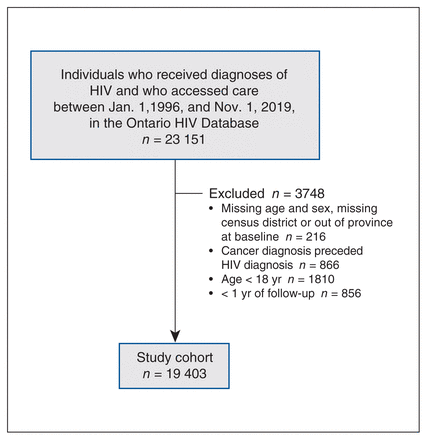
We identified adults (age ≥ 18 yr) with HIV using the Ontario HIV Database (OHIVD), an administrative registry of individuals with diagnosed HIV. We identified this population using a validated case-finding algorithm of 3 physician billing claims for an HIV-related visit within 3 years (codes 042–044 and B20–B24 in the 9th and 10th revisions of the International Classification of Diseases, respectively) in the Ontario Health Insurance Plan database (sensitivity 96.2%, specificity 99.6%).30 We excluded individuals who were missing information for age, sex or census district; who were out of province at cohort entry; who had less than 1 year of follow-up; or who had a cancer diagnosis before their diagnosis with HIV.
Cohort entry (baseline) was the earliest record of an HIV-related diagnostic code in the OHIVD case-finding algorithm. Individuals could enter the cohort from Jan. 1, 1996, to Nov. 1, 2019. Time at risk for incident cancers excluded the first 365 days of observation to avoid inclusion of cancers prevalent at HIV diagnosis.12,18,19 That is, we followed participants from 365 days after their cohort entry date to the earliest date of cancer diagnosis, date of death, loss to follow-up or Nov. 1, 2020. We defined loss to follow-up as no record of death and no contact with the health care system for at least 5 years; we censored individuals 5 years after the date of last contact.31–33
Data sources
We ascertained cancer diagnoses using the Ontario Cancer Registry, a passive registry containing data on incident cases of malignant neoplasia for all Ontario residents.34,35 We used the Canadian Institute for Health Information’s Discharge Abstract Database to identify hospital admissions and the Registered Persons Database (RPDB), a registry of all Ontario residents eligible for health insurance, for demographic and date of death data. We obtained neighbourhood-level income and rurality information based on postal code information. Finally, we used the Immigration, Refugees and Citizenship Canada Permanent Resident Database to identify immigrants to Ontario and their country of origin. The data sets were linked using unique encoded identifiers and analyzed at ICES.36–38
Outcome
The primary and secondary outcomes were incident and prevalent cancers, respectively. We classified first primary cancer sites using the International Agency for Research on Cancer’s (IARC) multiple primary coding rules39 and the International Classification of Diseases for Oncology, Third Edition.40 We grouped cancers as infection-unrelated and infection-related cancers, based on the IARC Group 1 biological agents classification.7 We further examined ADC and infection-related NADC separately to facilitate comparisons with other studies (Appendix 1, Supplemental Table 1A, available at www.cmajopen.ca/content/10/3/E666/suppl/DC1).
Distribution of baseline characteristics of adults living with HIV in Ontario, Canada, 1997–2020
Statistical analysis
We computed descriptive statistics for baseline continuous (median, interquartile range [IQR]) and categorical variables (n, %), including age, sex, rurality, neighbourhood income quintile, comorbidity burden, resource use, region of birth and immigration status. We used standardized differences to compare baseline characteristics between females and males, with a difference of less than 0.1 indicating good intergroup balance for a given variable.41 We used the Johns Hopkins Adjusted Clinical Group System Version 10 to measure comorbidity burden and to quantify expected use of health care resources into Resource Utilization Bands in the 2 years preceding cohort entry.42
We estimated age-standardized incidence rates by direct standardization, with corresponding 95% confidence intervals (CIs), using the gamma distribution method,43 and stratified them by calendar period and sex. We chose the 2011 Canadian population as the reference population given its use as a standard for provincial and national cancer estimates. 25,44,45 We used the Cochran–Armitage test of trend across calendar periods, using the χ2 statistic (with 1 degree of freedom) and p value. We conducted a sensitivity analysis excluding the COVID-19 pandemic months, from Jan. 1, 2020, to Nov. 1, 2020, and compared the prepandemic and peripandemic trends in incidence.
We estimated limited-duration prevalence for 2-, 5- and 10-year periods for individuals who were alive on Nov. 1, 2020, using the counting method,46 and stratified them by sex.
We performed analyses using SAS Enterprise Guide and created figures using STATA.
Ethics approval
The research ethics boards of St. Michael’s Hospital (No. 19-113), Toronto, and University of Toronto (No. 00038757) approved the study.
Results
Among 23 151 people diagnosed with HIV, 19 403 were included in the study (Figure 1). Most were male, born in Canada and lived in urban centres (Table 1). The median age at baseline was 36 (IQR 30–44) years. Compared with males, females were less likely to enter the cohort in the early combination ART era (1997–1999), more likely to live in the lowest neighbourhood income quintile, more likely to be born in a country with high HIV prevalence, more likely to have recently immigrated to Canada or immigrated after an HIV diagnosis, more likely to have a high comorbidity burden at baseline and more likely to have high resource use (Table 1).
Study flow chart.
There were 1275 incident first primary cancers over 193 527 person-years of observation (median 9 [IQR 4–15] yr), of which 631 (49.5%) and 644 (50.5%) were infection-unrelated and infection-related cancers, respectively. Most (n = 386, 60.0%) infection-related cancers were ADCs (Table 2). Compared with females, males had a higher proportion of ADCs and infection-related NADCs (Table 2). Of 239 cancers diagnosed among females, most were infection-unrelated cancers (n = 153, 64.0%).
Incident primary cancers diagnosed among people with HIV in Ontario, Canada, 1997–2020
From 1997 to 2020, non-Hodgkin lymphoma (19.1%), Kaposi sarcoma (12.6%), prostate (10.4%), lung (8.0%), anal (8.8%–9.4%) and colorectal cancer (5.9%) were the most common cancers among males, accounting for more than 65% of all cancers diagnosed among males (Appendix 1, Supplemental Table 2A). Among females, breast (18.0%), non-Hodgkin lymphoma (15.9%), lung (10.0%), thyroid (6.7%) and upper genital tract cancer (6.3%) were the most common cancers, constituting 57% of cancers diagnosed among females (Table 2). These trends persisted in the most recent calendar period (2016–2020), apart from cervical cancer displacing thyroid cancer from the top 5 most common cancers among females (data not shown).
Incidence trends
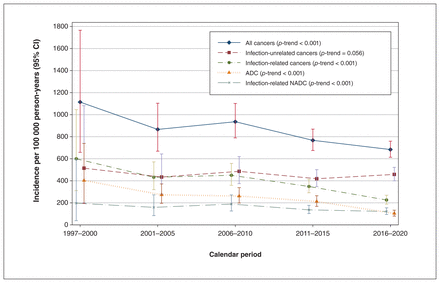
From the early combination ART era (1997–2000) to 2016–2020, the age-standardized incidence of cancer declined from 1113.9 (95% CI 657.7–1765.6) to 683.5 (95% CI 613.4–759.4) cases per 100 000 person-years (p-trend < 0.001) (Figure 2; Appendix 1, Supplemental Table 3A). Similarly, the age-standardized incidence of infection-related cancer declined from 599.7 (95% CI 310.7–1045.6) to 225.6 (95% CI 188.0–268.6) per 100 000 person-years (p-trend < 0.001). The slope of the age-standardized rate of infection-unrelated cancers was relatively stable (p-trend 0.056; 451.0 per 100 000 person-years, 95% CI 410.3–494.7) (Figure 2), although the crude rate increased from 255.1 to 393.3 per 100 000 person-years (Appendix 1, Supplemental Figure 1A).
Age-standardized incidence per 100 000 person-years of first primary cancers among people with HIV, by calendar period and cancer category. Note: ADC = AIDS-defining cancer, CI = confidence interval, NADC = non-AIDS-defining cancer.
Two-, 5- and 10-year person-based prevalence of cancer as of end of follow-up (Nov. 1, 2020)
From 1997–2000 to 2016–2020, we observed declines in the age-standardized incidence of ADC, from 403.1 (95% CI 194.2–739.0) to 103.8 (95% CI 79.2–133.6) cases per 100 000 person-years (p-trend < 0.001), and of infection-related NADC, from 196.6 (95% CI 37.9–591.9) to 121.9 (95% CI 94.3–154.9) per 100 000 person-years (p-trend < 0.001) (Figure 2). In contrast, the crude rate increased between 1997 and 2015 (Appendix 1, Supplemental Figure 1A). The sensitivity analysis excluding the COVID-19 pandemic year showed that the incidence trends remained the same (data not shown).
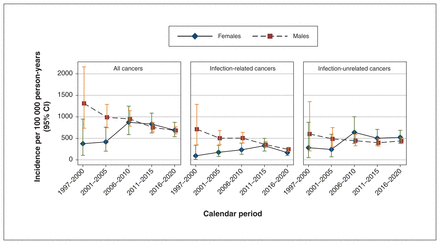
Among males, the age-standardized incidence of cancer decreased over time from 1311.4 (95% CI 735.2–2160.5) to 681.8 (95% CI 603.7–767.2) per 100 000 person-years (p-trend < 0.001) (Figure 3; Appendix 1, Supplemental Table 4A). This was largely driven by the considerable decline in infection-related cancers (from 711.2 [95% CI 347.9–1290.3] to 241.3 [95% CI 197.8–291.5] per 100 000 person-years; p-trend < 0.001).
Age-standardized incidence per 100 000 person-years of first primary cancers among people with HIV, by calendar period and sex. Note: CI = confidence interval.
Among females, the age-standardized incidence of cancer increased from 375.8 (95% CI 105.3–948.6) to 690.5 (95% CI 538.9–871.4) per 100 000 person-years (p-trend < 0.001) (Figure 3; Appendix 1, Supplemental Table 4A). The largest increase occurred for infection-unrelated cancers, which increased from 281.6 (95% CI 50.8–872.4) to 523.4 (95% CI 390.8–686.6) cases per 100 000 person-years (p-trend < 0.001).
Crude incidence rates of the top 5 most common cancers among males and females are included in a supplemental analysis (Appendix 1, Table 5A). We were unable to estimate age-standardized sex-specific rates because of small cell sizes.
Prevalence
Among the 17 181 people alive on Nov. 1, 2020, 0.8% (n = 139), 1.8% (n = 313) and 3.0% (n = 510) had a cancer diagnosis in the past 2, 5 and 10 years, respectively (Table 3). Prevalence proportions were highest for infection-unrelated cancers, followed by infection-related cancers (Table 3). There were no significant differences in 2-, 5- and 10-year prevalence proportions between males and females (data not shown).
Interpretation
In this large, population-based cohort in Ontario, Canada, more than 1000 incident cancers occurred among people with HIV over more than 190 000 person-years of follow-up. Prostate, lung, anal (HPV-related), Kaposi sarcoma and non-Hodgkin lymphoma accounted for 54% of all cancers in people with HIV. These findings are congruent with modelling projections of lung and prostate cancers being the most common cancer types among people with HIV by 2030.47
Consistent with previous Canadian and other North American studies, we observed a shift in cancer burden from predominantly ADCs to NADCs, corresponding to the period of modern ART.5,6,10–13,15,20,21,48 We detected a downward trend in the age-adjusted overall incidence of cancer from 1997 to 2020, driven mainly by the considerable decrease in ADC incidence. Incidence rates of infection-unrelated cancer were stable over time, whereas we saw a decline in infection-related NADC, which slowed between 2011–2015 and 2016–2020. Moreover, the incidence of infection-unrelated cancer was about 2 times higher than infection-related NADC across the study period. Finally, we identified an increasing trend in cancer incidence among females, driven by the rise in incidence of infection-unrelated cancers, whereas cancer incidence among males decreased. As expected, given the aging HIV population in Ontario,49 crude incidence rates increased for both infection-related and infection-unrelated NADCs.
Although not directly explored in this study, our estimates of cancer incidence rates in people with HIV in Ontario exceed reported rates for the general populations of Ontario and Canada by about 1.2-fold.25,45 Our estimates of cancer incidence were likely an underestimate given the known disparities in cancer screening in Ontario between people with HIV and the general population.50–52 Factors that likely contribute to the higher rates of cancer among people with HIV are differences in rates of smoking, alcohol use and the role of chronic inflammation and immune suppression.7–9
The years of study largely excluded the COVID-19 pandemic, and our sensitivity analysis did not show a difference in trends with exclusion of follow-up from March to November 2020. Nonetheless, others have found reduced rates of cancer screening and diagnoses53 and fewer primary care visits54 during the pandemic, suggesting that ongoing monitoring is needed.
As of the end of 2020, we found that 3% of people living with HIV in Ontario were cancer survivors. Prevalence estimates are crucial to understanding the nature of health care and support services that may improve the quality of life for people with HIV and cancer as they transition through different stages of diagnosis, treatment and recovery from cancer. Of note, nearly 1% and 2% of people had a cancer diagnosis in the previous 2 and 5 years, respectively, and likely required cancer care and close clinical follow-up for recurrence.
Our study provides a unique perspective in a setting with publicly funded health care. Strengths include the use of comprehensive, high-quality administrative and cancer registry data. Given the long follow-up period, we were able to capture many cancer cases and further examine infection-related and infection-unrelated cancers, as well as describe sex-specific trends.
Limitations
Among people lost to-follow-up, assumptions regarding the end of follow-up time may have resulted in overestimation of person-time denominators. Despite our large cohort, low site-specific cancer counts meant that we had to aggregate cancers into larger groups and estimate incidence over 5-year periods rather than annually. Our use of a validated algorithm to identify people with HIV in health administrative data necessarily excluded individuals with undiagnosed HIV and those who did not access care. We also lacked information on known cancer risk factors, including lifestyle factors (e.g., smoking, alcohol) and clinical HIV data (e.g., CD4, coinfection with hepatitis B or C, combination ART), precluding their exploration.3,9,55
Conclusion
Despite the substantial decline in ADC incidence, there was little to no decrease in the incidence of infection-related NADC and infection-unrelated cancer over the study period. The prevalence and incidence of cancer were also considerable, compared with the general populations of Ontario and Canada. Consequently, it is important for clinicians to incorporate cancer prevention strategies as part of comprehensive HIV care. This includes discussing cancer risk with patients, encouraging risk reduction such as smoking cessation, and encouraging uptake of primary and secondary interventions for cancer prevention, such as HPV vaccination, hepatitis C treatment and screening for HPV-related cancers and colorectal cancer.
Acknowledgements
In memoriam of Zak Knowles, formerly at CATIE Toronto Ontario, Canada. Zak Knowles was a coinvestigator on the grant (No. EFP-1104-GC) that supported this study and made substantial contributions to conception and design, analysis and interpretation of data. He passed away before the submission of this article. His contributions will be deeply missed by the HIV community. The authors thank the community engagement committee members, as well as their community partner agencies. The authors thank Karl Everett at ICES Central for his contributions to data preparation.
Footnotes
Competing interests: Marek Smieja reports funding from the Canadian Institutes of Health Research, Air Canada and the Greater Toronto Airports Authority. No other competing interests were declared.
This article has been peer reviewed.
Contributors: Ann N. Burchell and Ioana Nicolau were involved in the study concept and design. Ioana Nicolau conducted the analysis with supervision from Ann N. Burchell, Tony Antoniou, Jennifer D. Brooks and Rahim Moineddin. All authors interpreted the data. Ioana Nicolau drafted the manuscript. All authors revised it critically for important intellectual content, approved the final version to be published and agreed to be accountable for all aspects of the work.
Funding: This study received funding from Ontario HIV Treatment Network Endgame Game Changer Grant (No. EFP-1104-GC). Ioana Nicolau was funded by a Canadian Institutes of Health Research Doctoral Award (No. 201710MDR) and the Canadian HIV Observational Cohort Scholar Award (No. 711352). Ann Burchell is a Canada Research Chair in Sexually Transmitted Infection Prevention (Tier 2). During the study, Ann Burchell received support from an Ontario HIV Treatment Network (OHTN) Endgame Leader award and a Non-Clinician Researcher award from the Department of Family and Community Medicine, Faculty of Medicine, University of Toronto. Jennifer Gillis received support from the OHTN Endgame Student Leadership Award. Claire Kendall was supported by CIHR New Investigator award and Clinical Research Chair Award from the Faculty of Medicine University of Ottawa.
Data sharing: The data set from this study is held securely in coded form at ICES. While legal data sharing agreements between ICES and data providers (e.g., health care organizations and government) prohibit ICES from making the data set publicly available, access may be granted to those who meet prespecified criteria for confidential access, available at https://www.ices.on.ca/DAS (email: das{at}ices.on.ca). The full data set creation plan and underlying analytic code are available from the authors upon request, understanding that the computer programs may rely upon coding templates or macros that are unique to ICES and are therefore either inaccessible or may require modification.
Disclaimer: Parts of this material are based on data and/or information compiled and provided by the Canadian Institute for Health Information; Immigration, Refugees and Citizenship Canada (IRCC), current to March 2020; Ontario Health (Cancer Care Ontario). The analyses, results, conclusions, opinion, views and statements expressed herein are solely those of the authors and do not reflect those of the funding and data sources; no endorsement is intended or should be inferred. This study was supported by ICES, which is funded by an annual grant from the Ontario Ministry of Health (MOH) and the Ministry of Long-Term Care (MLTC). The opinions, results and conclusions reported in this article are those of the authors and are independent from the funding sources. No endorsement by ICES or the Ontario MOH and MLTC is intended or should be inferred.
Supplemental information: For reviewer comments and the original submission of this manuscript, please see www.cmajopen.ca/content/10/3/E666/suppl/DC1.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/
References
- © 2022 CMA Impact Inc. or its licensors