Abstract
Background: Information about the timing involved in various stages of making new drugs available to Canadians is important for understanding how a national pharmacare plan will affect timely access to new drugs. I explored the timing of the various steps between receiving a Notice of Compliance and a decision by the pan-Canadian Pharmaceutical Alliance (pCPA).
Methods: I gathered data from various databases (Canadian and other) about new drugs approved between 2011 and 2020, including generic names, date of application for approval (New Drug Submission [NDS]), date of Notice of Compliance, date of marketing, dates when a submission was made to the Canadian Agency for Drugs and Technologies in Health (CADTH) and the pCPA, and when these agencies made a decision.
Results: Marketing dates were available for 301 of the 337 new drugs approved. The median time from NDS to marketing was less than the time to a positive pCPA decision for all years between 2011 and 2020. There was no significant change in the difference between the 2 periods over time (p = 0.2). Additional therapeutic value did not make a difference in the delay (p = 0.3) and companies did not take full advantage of the opportunity to file early submissions with CADTH.
Interpretation: The delay between when drugs could be listed on private compared with public formularies was at least 1 year. If a national pharmacare plan is instituted, one of the priorities should be to concentrate on consolidating and working to shorten the CADTH and pCPA processes.
Making new active substances (drugs that have not been marketed in Canada in any form) available to patients is a complex process with a number of steps. Once the clinical trials have been completed, companies file a New Drug Submission (NDS) with Health Canada to initiate the approval process. If the evidence of safety, efficacy and manufacturing quality is acceptable, the product is then approved (i.e., issued a Notice of Compliance [NOC]).1 Companies are then able to market their product and it can be listed on private insurance plans that cover 60% of Canadians.2 If companies want to have the cost of the drug covered by a public drug plan, they would file an application with the Canadian Agency for Drugs and Technologies in Health ([CADTH], a third-party, government-funded, health technology assessment agency).3 In Quebec, the health technology assessment is conducted by the Institut national d’excellence en santé et en services sociaux [INESS].4 Since 2012, companies have had the option to submit to CADTH before an NOC had been issued.5 Once CADTH has finished the assessment, a recommendation to public drug plans is then issued. After CADTH makes a recommendation, the public drug plans may jointly negotiate a price with the manufacturer through the pan-Canadian Pharmaceutical Alliance (pCPA).6 If negotiations are successful, public drug plans have the option of listing the drug on their formularies and it is then available to patients covered by these plans.
Consumer and patient groups,7,8 the pharmaceutical industry8,9 and private insurers8,10 have expressed concerns about the time difference in access between private and public insurance plans, with regards to delay in access if a national pharmacare plan is instituted.
Some of the time periods have been previously investigated. Lexchin reported on the median time between NOC and marketing.11 Dobrescu gave the time between filing a CADTH application and an NOC and from an NOC to a first pCPA decision.9 Salek and colleagues analyzed the time it took between submissions to CADTH, its recommendations and the similar time for pCPA.5 Finally, Rovere and Skinner12 stated that private plans take 4 months to add medicines to their formularies, compared with 16 months for public plans.
Examining the timing of the various steps is essential for understanding the delay in listing between public and private formularies and for how a national pharmacare plan may affect the time it will take for people to access medications. I explored key time periods between approving drugs and listing to private and public formularies for all drugs and drugs of different therapeutic value:
What is the difference in length of time between when drugs are marketed and can be listed on private insurance plans and when a pCPA decision is delivered, making drugs potentially available for listing on public plans? Has that difference changed between 2011 and 2020?
Does the additional therapeutic value of a drug compared with already existing drugs affect the difference as defined in the first question?
What is the time between when a drug is approved and when it receives a positive pCPA decision, and has that time changed between 2011 and 2020?
Starting in 2012, CADTH accepted submissions from companies up to 90 days before an NOC and that was increased to 180 days from Apr. 2, 2018, onward. According to Heather Logan, vice-president (acting) of pharmaceutical reviews (CADTH), these changes were made to eliminate delays between regulatory approval and CADTH funding recommendations.5,13 Have pharmaceutical companies used this opportunity to accelerate CADTH recommendations?
Methods
I conducted a cross-sectional study looking at the timing of various regulatory decisions and recommendations for drugs approved by Health Canada.
Data sources
A list of all drugs approved in Canada between Jan. 1, 2011, and Dec. 31, 2020, was compiled from the annual reports of the Therapeutic Products Directorate and the Biologic and Radiotherapeutic Drugs Directorate (reports are available by contacting the directorates at publications{at}hc-sc.gc.ca). The generic drug names were recorded in a spreadsheet along with the dates of when the companies filed an NDS and the drug received an NOC.
Health Canada’s Drug Product Database14 supplies the date of when a product is originally marketed, and is defined on the website as the “earliest marketed date recorded in the Drug Product Database,” but no further information is provided about how that date is determined. The marketing date was recorded on the same spreadsheet.
CADTH makes the timelines for the drugs that it reviews publicly available (https://www.cadth.ca/reimbursement-review-reports). The dates of the submission and final recommendation were recorded, along with the type of recommendation (fund, fund with conditions and do not fund). Previous to March 2016, the categories were as follows: do not list, do not list at the submitted price, list with clinical criteria or conditions and list.15 I collapsed into a single category the pre-March 2016 categories of do not list and do not list at the submitted price. If more than 1 submission for a single drug was made, only the dates of the initial submission and recommendation and the type of recommendation were used. Similarly, the pCPA website (https://www.pcpacanada.ca/negotiations) gives the date of when it issues its decision along with the results of the negotiations (positive, negative, negotiations not pursued, withdrawn and under consideration for negotiation). If the pCPA conducted more than 1 negotiation for the same product, the date of a positive outcome was used. The data from both CADTH and the pCPA were recorded in the same spreadsheet as the other data.
To confirm the accuracy of the data, all databases were searched between Feb. 5 and 15, 2022, and a second full data abstraction was done between June 18 and 20, 2022. INESS decisions and the time of listing on federal, provincial or territorial formularies were not recorded. All extracted data are available in Appendix 1, available at www.cmajopen.ca/content/10/4/E993/suppl/DC1.
Therapeutic evaluation
I assessed the additional therapeutic value of drugs compared with existing drugs by consulting the evaluations conducted by the Human Drug Advisory Panel of the Patented Medicine Prices Review Board (http://www.pmprb-cepmb.gc.ca/pmpMedicines.asp?x%20=%20611) and Prescrire International, an independent French drug bulletin (https://english.prescrire.org/en/Summary.aspx). Therapeutic value was graded as major, moderate or little to no additional therapeutic value. Table 1 shows how the ratings of the 2 organizations were equated. These 2 organizations were chosen because they used ordinal ratings for therapeutic value, they reported in English and their ratings had been accepted in other research.16 Briefly, the Human Drug Advisory Panel considered 2 primary factors: increased efficacy and reduction in incidence or grade of important adverse reactions and 9 secondary factors with the primary factors being given the greatest weight. Prescrire International assessed the therapeutic value of medicines through a multistep process including a systematic search for clinical data on the efficacy and adverse effects of the new drug and an assessment of the level of evidence followed by peer review of its analysis.17
Evaluation of therapeutic value
Data analysis
I based analyses on the year that the drug received an NOC. Medians and interquartile ranges (IQRs) were used instead of means as the data were not normally distributed.
Time differences were calculated in days for each drug and grouped by year for each year between 2011 and 2020. The medians were compared by the time between submission of the NDS and the marketing date and the time between the NDS submission date and the date of a pCPA decision. The time between when a drug received an NOC and when a pCPA decision was made was also calculated and the interval was compared for each year between 2011 and 2020. I calculated for the entire time period and compared the difference between the median time to market and time to a pCPA decision for drugs with 3 different therapeutic ratings.
The time in days between the date when the CADTH submission was filed and when an NOC was issued was calculated for all drugs with a CADTH submission, except for 1 product for which the submission was initiated by drug plans and not the manufacturer. The difference in median times were compared for drugs approved in 3 time periods: 2011, before companies had the opportunity to submit a pre-NOC application to CADTH; 2012 to Apr. 1, 2018, when companies could submit 90 days before an NOC; and Apr. 2, 2018, to Dec. 31, 2020, when companies could submit 180 days before an NOC.
All calculations were completed with Prism 9.3.1 (Graph-Pad Software, LLC). I used the Kruskal–Wallis test with a 2-sided p value of less than 0.05 to assess the significance of the difference across years and therapeutic value.
Ethics approval
All data were publicly available and ethics approval was not deemed necessary.
Results
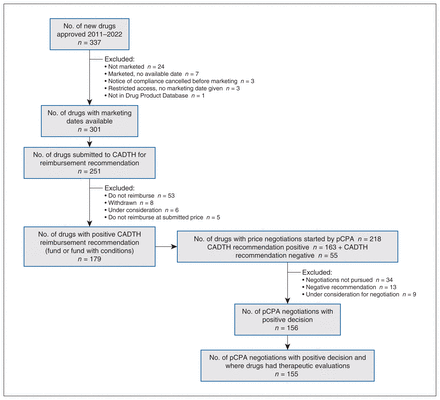
From Jan. 1, 2011, to Dec. 31, 2020, Health Canada approved 337 drugs, with marketing dates available for 301. There were 251 submissions to CADTH, which issued 179 positive recommendations (13 fund and 166 fund with conditions). There were 218 submissions to the pCPA, with 156 successfully completed (138 had a positive CADTH recommendation and 18 had a negative recommendation). From the 156 submissions, 155 drugs had a therapeutic evaluation. Of note, pCPA conducted negotiations for 163 drugs with a positive CADTH recommendation and 55 with a negative CADTH recommendation. Figure 1 shows the relation between the number of drugs at the different stages of the process.
Number of new drugs at various stages of the process of potential formulary listing. Note: CADTH = Canadian Agency for Drugs and Technologies in Health, pCPA = pan-Canadian Pharmaceutical Alliance.
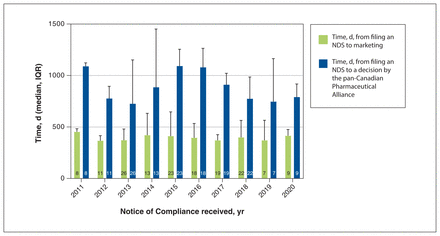
The time from NDS submission to marketing was less than the time from NDS submission to a pCPA outcome from 2011 to 2020 (Figure 2). The annual median differences ranged from 354 (IQR 202 to 572) days in 2018 to 650 (IQR 403 to 753) days in 2016 (Figure 3); however, the difference in the delay between years was not significant (p = 0.2).
Time in days from filing an NDS to marketing and to a decision from the pan-Canadian Pharmaceutical Alliance. Note: Column numbers are the number of drugs evaluated, IQR = interquartile range, NDS = New Drug Submission.
Difference in days between marketing data and pan-Canadian Pharmaceutical Alliance decision. Note: Column numbers are the number of drugs evaluated, IQR = interquartile range.
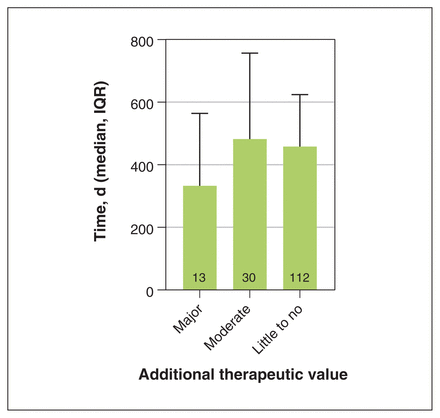
For the 10-year time period, the median difference in time between marketing and a positive pCPA outcome was 333 (IQR 290 to 564) days, 482 (IQR 308 to 757) days and 459 (IQR 311 to 625) days for drugs with major, moderate and little to no additional therapeutic value, respectively (Figure 4). There was no significant difference in the delays (p = 0.3).
Difference in days between marketing date and receiving a positive pan-Canadian Pharmaceutical Alliance decision as a function of additional therapeutic value. Note: Column numbers are the number of drugs evaluated, IQR = interquartile range.
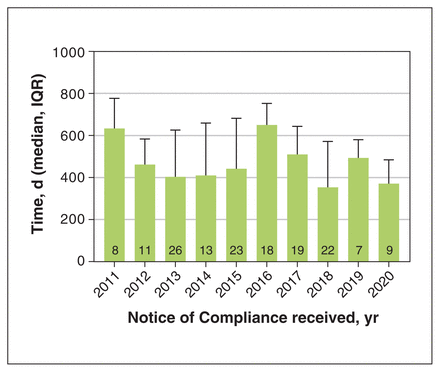
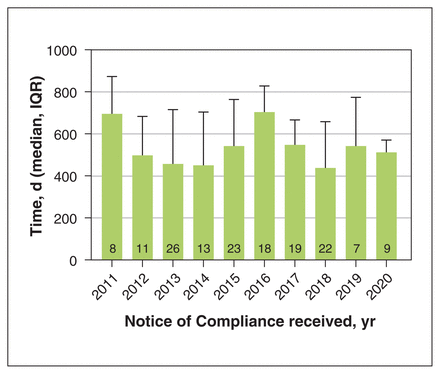
The time between an NOC and a pCPA decision ranged from 438 (IQR 341 to 658) days in 2018 to 704 (IQR 548 to 828) days in 2016 (Figure 5). There was no significant difference in the delay (p = 0.2).
Number of days between a Notice of Compliance and pan-Canadian Pharmaceutical Alliance decision. Note: Column numbers are the number of drugs evaluated, IQR = interquartile range.
In 2011, companies filed CADTH submissions 66 (IQR 7 to 218) days after a drug received an NOC. From Jan. 1, 2012, to Apr. 1, 2018, the difference was 4 (IQR −71 to 78) days and from Apr. 2, 2018, to Dec. 31, 2020, the CADTH submission was filed 13 (IQR −136 to 139) days before the NOC. The difference between periods was not significant (p = 0.08). Table 2 summarizes the various time intervals.
Difference in times between Notice of Compliance and pan-Canadian Pharmaceutical Alliance decision
Interpretation
I found that between 2011 and 2020, the time between when a drug was marketed and when it received a pCPA decision ranged from a median of 354 days to 650 days, although these intervals were not significantly different. These differences did not take into account the time taken for listing on a public formulary, which Salek and colleagues4 reported as a mean of 67 days for drugs evaluated by CADTH between 2012 and 2016. Depending on the year, the overall difference between private and public availability was about 12 months as reported by Rovere and Skinner.12
There was no significant difference in the time between marketing and a positive pCPA decision for drugs with different additional therapeutic values. This finding contrasts with company behaviour for drugs with an expedited regulatory review (priority or NOC with conditions review) compared with a standard regulatory review. In that case, companies were quicker to market drugs with the former type of review than the latter.11
There was no change in the time between when a drug received an NOC and when the pCPA rendered a decision over the 10-year period. These results show there was neither a deterioration nor an improvement in the overall process used to list public drug plans. The graphical results reported by Dobrescu showed a lengthening of this period,9 as the time in 2013 was 404 days compared with 568 days in 2020. However, it is not clear if means or medians were reported and there was no statistical analysis of the differences.
Companies did not appear to be taking full advantage of the opportunity to submit most of their drugs for CADTH evaluation before the NOC. From 2012 to Apr. 1, 2018, they could have submitted applications 90 days before an NOC, whereas the median time was 4 days after an NOC. After Apr. 2, 2018, companies could have filed a submission 180 days before an NOC but they were doing so only 13 days before. These results are broadly in line with what Dobrescu found, although this analysis did not include a statistical comparison to see if time periods were changing.9 Had companies taken full advantage of the opportunity, drugs might have been available for listing on public formularies much earlier. Companies may not have been filing earlier because they were uncertain if their products would receive an NOC and they did not want to expend resources unnecessarily. Another possibility is that a negative recommendation from CADTH could affect decisions by other countries that use health technology assessments.
Limitations
My results apply only to the specific subsets of drugs for which data were available. There were only pCPA decisions for 156 of the 337 drugs that were approved; therefore, I cannot draw any conclusions about whether the remaining 181 were eligible for public funding and, if they were, what the interval between private and public formulary listing may have been. I used a secondary analysis of a number of Canadian government databases. Those databases were assembled and published based on information either provided to, or generated by, the federal government and are regarded as authoritative. No formal evaluation of their quality or validity was conducted. Although the data do not require any subjective interpretation, there was the possibility for data entry errors and missing data; for example, drug approvals may be missing from annual reports. There was the assumption that the evaluations by the Patented Medicine Prices Review Board or Prescrire International represented a gold standard in the assessment of a drug’s therapeutic gain. Although there was a legitimate debate about therapeutic gain, the rigorous processes that these organizations used and their independence gave strong face validity to their assessments. Finally, the data were gathered and analyzed by a single person.
Conclusion
There was at least a 1-year interval between when drugs could be listed on private compared with public formularies. This interval did not depend on the additional therapeutic value of the drugs compared with existing drugs. The difference is important to patients for drugs that offer the greatest therapeutic improvement. Both companies and the various regulatory organizations should concentrate on moving these drugs through the various processes more rapidly. Companies could expedite public availability by taking full advantage of their ability to file submissions with CADTH before receiving an NOC. If a national pharmacare plan is initiated, one of the priorities should be to shorten the CADTH and pCPA processes. Changes to the CADTH and pCPA processes could be the subject for a separate article.
Acknowledgement
Oliver Spicer made valuable comments on an earlier version of this manuscript.
Footnotes
Competing interests: Joel Lexchin received payments for writing a brief in an action for side effects of a drug for Michael F. Smith, lawyer, and a second brief on the role of promotion in generating prescriptions for Goodmans LLP, in 2018–2021. He is a member of the Foundation Board of Health Action International. He receives royalties from the University of Toronto Press and James Lorimer & Co. Ltd. for books he has written. He has received payments from the Canadian Institutes of Health Research for presenting at a workshop on conflict of interest in clinical practice guidelines.
This article has been peer reviewed.
Contributors: Joel Lexchin conceived and designed the work and acquired and analyzed the data. He interpreted the data, revised the manuscript critically for important intellectual content, gave final approval of the version to be published and agreed to be accountable for all aspects of the work.
Data sharing: All data collected for this study are available in Appendix 1.
Supplemental information: For reviewer comments and the original submission of this manuscript, please see www.cmajopen.ca/content/10/4/E993/suppl/DC1.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/
References
- © 2022 CMA Impact Inc. or its licensors