Abstract
Background: People with HIV infection are at higher risk for certain cancers than the general population. We compared trends in infection-related and infection-unrelated cancers among people with and without HIV infection.
Methods: We conducted a retrospective population-based matched cohort study of adults with and without HIV infection using linked health administrative databases in Ontario, Canada. Participants were matched on birth year, sex, census division (rurality), neighbourhood income quintile and region of birth. We followed participants from cohort entry until the earliest of date of cancer diagnosis, date of death, Nov. 1, 2020, or date of loss to follow-up. Incident cancers identified from Jan. 1, 1996, to Nov. 1, 2020, were categorized as infection-related or-unrelated. We examined calendar periods 1996–2003, 2004–2011 and 2012–2020, corresponding to the early combination antiretroviral therapy (cART), established cART and contemporary cART eras, respectively. We used competing risk analyses to examine trends in cumulative incidence by calendar period, age and sex, and cause-specific hazard ratios (HRs).
Results: We matched 20 304 people with HIV infection to 20 304 people without HIV infection. A total of 2437 cancers were diagnosed, 1534 (62.9%) among infected people and 903 (37.0%) among uninfected people. The risk of infection-related cancer by age 65 years for people with HIV infection decreased from 19.0% (95% confidence interval [CI] 15.6%–22.3%) in 1996–2011 to 10.0% (95% CI 7.9%–12.1%) in 2012–2020. Compared to uninfected people, those with HIV infection had similar HRs of infection-unrelated cancer but increased rates of infection-related cancer, particularly among younger age groups (25.1 [95% CI 13.2–47.4] v. 1.9 [95% CI 1.0–3.7] for age 18–39 yr v. ≥ 70 yr); these trends were consistent when examined by sex.
Interpretation: We observed significantly higher rates of infection-related, but not infection-unrelated, cancer among people with HIV infection than among uninfected people. The elevated rate of infection-related cancer in 2012–2020 highlights the importance of early and sustained antiretroviral therapy along with cancer screening and prevention measures.
People with HIV infection receiving combination antiretroviral therapy (cART) have, on average, twice the risk of cancer as the general population.1–3 Before the introduction of cART, in 1996, the most common cancers among people with HIV infection were Kaposi sarcoma, non-Hodgkin lymphoma and invasive cervical cancer, classified as AIDS-defining cancers (ADCs).2,4,5 In recent years, non–AIDS defining cancers (NADCs) have become increasingly common2,4,5 given the success of cART, bringing life expectancy near that of the general population.6 Cancer risks reflect the interaction of multiple component causes, including HIV-related immunosuppression and decreased immune surveillance of oncogenic infections, and a higher prevalence of known cancer risk factors such as tobacco and alcohol use, obesity, viral hepatitis and human papillomavirus (HPV) infection.3,7,8
Most studies to date comparing cancer incidence among people with and without HIV infection were conducted in Europe and the United States,1–5,9–17 with comparatively few studies performed in Canada.18–20 A recent meta-analysis of 47 independent studies from the Americas, Europe, Africa and Asia, with follow-up from 1978 to 2010, showed an increased standardized incidence ratio for all 20 infection-related NADCs identified and half of the 20 infection-unrelated cancers among people with HIV infection compared to the general population.17 There was moderate to high between-study heterogeneity for almost all outcomes, which underlines the need for population- and time-specific estimates. Moreover, context-specific current data are needed to guide medical care and clinical decision-making at the community level because cancer epidemiologic features in one HIV population may not apply in another setting: there are differences in the local epidemiologic features of HIV acquisition, patterns of coinfection with other oncogenic agents and prevalence of cancer risk factors. Given that these factors vary substantially across HIV populations and jurisdictions, it is important to add to the current evidence in the Canadian context.
Among the Canadian provinces and territories, Ontario has the highest number of annual new HIV infections and the largest population of people living with HIV infection.21,22 We previously documented a substantial decline in ADC incidence, from 403 to 104 per 100 000 person-years, between 1997 and 2020 in Ontario, but little to no decrease in the incidence of infection-unrelated cancer.23 Our objective in the present study was to extend this work to compare calendar and age-related trends in the incidence of infection-related and infection-unrelated cancers among people with HIV infection to those in a matched cohort of people without HIV infection, from 1996 to 2020.
Methods
Study design and setting
We conducted a retrospective population-based matched cohort study in Ontario from Jan. 1, 1996, to Nov. 1, 2020, using linked health administrative data sets housed at ICES. ICES is an independent, nonprofit research institute whose legal status under Ontario’s health information privacy law allows it to collect and analyze health care and demographic data, without consent, for health system evaluation and improvement. We used the Reporting of Studies Conducted Using Observational Routinely Collected Health Data (RECORD) and Guidance for Reporting Involvement of Patients and the Public Short Form (GRIPP2-SF) reporting checklists.24,25
As part of our patient and public engagement initiatives, we involved community members and people living with both HIV infection and cancer to guide research development. Two community members (C.P. and J.D.L.) were involved as coinvestigators, providing support and feedback during study development and throughout the research process, and as coauthors. We also established a community advisory board to provide the research team with ongoing feedback and to contextualize findings within members’ lived experience. The researchers were experienced at involving community partners in their research and community engagement from study inception, which allowed community partners to influence aspects of the study early in the study development.
Participants
We identified Ontario residents aged 18 years or older with a diagnosed HIV infection according to a previously described validated case-finding algorithm.23,26 We defined cohort entry as the earliest record of an HIV-related diagnostic code in the case-finding algorithm. We excluded people who were missing age or sex information, who at cohort entry were not residing in Ontario, or who had a previous cancer diagnosis. The look-back periods for cancer diagnosis and HIV diagnosis were 1964–2020 and 1991–2020, respectively. Each person with HIV infection was matched 1:1 with a person without a diagnosis of HIV as of the cohort entry date (within 1 yr). People were matched on birth year (within 1 yr), sex, census division (rurality), neighbourhood income quintile and region of birth (Canadian born or immigrated to Canada before 1986, born outside Canada in a country with low HIV infection prevalence, or born outside Canada in a region with high HIV infection prevalence).27 We followed participants from cohort entry until the earliest of date of cancer diagnosis, date of death, Nov. 1, 2020, or date of loss to follow-up, defined as no record of death and no contact with the health care system for at least 5 years; we censored individuals 5 years after the date of last contact.28–31
Data sources
We ascertained cancer diagnoses from the Ontario Cancer Registry, which records information for all primary incident cancers (except for basal cell and squamous cell skin cancers) using the International Classification of Diseases for Oncology, Third Edition.32,33 We used the Registered Persons Database, a registry of all Ontario residents with health insurance, for demographic and date of death information. We used the Canadian Institute for Health Information’s Discharge Abstract Database and the Ontario Health Insurance Plan database to identify hospital admissions, claims for physician services and comorbidity burden. We used the Immigration, Refugees and Citizenship Canada Permanent Residents Database to identify immigration status and region of birth. Finally, we used postal code information to obtain neighbourhood-level income and rurality. The data sets were linked by means of unique encoded identifiers and analyzed at ICES.34–36
Outcomes and covariates
Our primary outcomes were infection-unrelated and infection-related cancers (Box 1; Appendix 1, Supplementary Table S1, available at www.cmajopen.ca/content/11/5/E894/suppl/DC1).37 In secondary analyses, we examined the incidence of ADCs, infection-related NADCs and the most commonly observed NADCs in our cohort.
List of infection-related and infection-unrelated cancers37
Infection-related cancers
Kaposi sarcoma
Non-Hodgkin lymphoma
Cervical cancer
Anogenital cancers (including anus, penis, unspecified male genital organs,* vulva, vagina, oral cavity and pharynx)
Stomach
Liver
Nasopharynx
Hodgkin lymphoma
Infection-unrelated cancers
Bladder
Brain
Breast
Colorectal
Esophagus
Kidney
Larynx†
Leukemia
Lung
Melanoma of the skin
Multiple myeloma
Ovary
Pancreas
Prostate
Testis
Thyroid
Uterus
We ascertained the following information on covariates at cohort entry: age, sex, rurality, neighbourhood income quintile, region of birth, immigration status, comorbidity based on type of illness according to the Johns Hopkins Adjusted Clinical Group System and diagnosis codes in the 2 years preceding cohort entry,39,40 and expected use of health care resources in the 2 years prior, assessed with the Johns Hopkins Adjusted Clinical Groups Resource Utilization Bands based on morbidity level (not type of illness) and classified into 6 categories from 0 (no health care use) to 5 (highest expected use).40 We examined calendar periods 1996–2003, 2004–2011 and 2012–2020, corresponding to the early cART, established cART and contemporary cART eras, respectively.41,42 We classified attained age (time-varying) as early adulthood (18–39 yr), early and late middle adulthood (40–49 yr and 50–59 yr, respectively), and early and late old adulthood (60–69 yr and ≥ 70 yr, respectively). 43–47 CD4 status, viral load and antiretroviral information were unavailable.
Statistical analysis
We used descriptive statistics to summarize categoric (frequency and proportion) and continuous (median and interquartile range [IQR]) variables at cohort entry. We used standardized differences to compare characteristics at cohort entry between people with and without HIV infection, with a difference of less than 0.1 indicating good intergroup balance.48
We plotted unadjusted nonparametric cumulative incidence curves for the primary and secondary outcomes, and estimated cumulative cancer risk by age 65 years and age 75 years.11 We used cause-specific hazards models to compare cancer incidence between people with and without HIV infection, stratified by calendar period, attained age and sex (for primary outcomes only), with all-cause mortality as a competing event and censoring at loss to follow-up and end of follow-up.
When examining infection-related cancers, we considered infection-unrelated cancers as a competing event in addition to all-cause mortality since, by definition, being diagnosed with an infection-unrelated first primary cancer precludes diagnosis of an infection-related first primary cancer. Similarly, for infection-unrelated cancers, we considered infection-related cancer as a competing risk.
We carried out the cause-specific model by implementing the Cox proportional hazards model and censoring at competing events. Using the Cox proportional hazards model, we obtained cause-specific hazard ratios (HRs) to estimate the relative effect of HIV status on the hazard function.49 We estimated 95% confidence intervals (CIs) using the robust sandwich covariance estimate.50 We adjusted all models for comorbidity, expected resource use and immigration status. Rurality, region of birth and neighbourhood income quintile were not included in the regression models because we matched on these covariates.51,52 We assessed the proportionality of hazards assumption using interaction terms between HIV status and time and weighted Schoenfeld residuals. In sensitivity analysis, we calculated the E-value to examine the amount of confounding bias needed to completely explain away the observed HRs.53,54
We used the RStudio package “prodlim,” version 0.98.1091 to estimate cumulative incidence functions and to plot cumulative incidence curves. We conducted all other analyses, including estimates of adjusted cause-specific HRs, using SAS Enterprise Guide version 7.15 (SAS Institute). We used the SAS macro %match for the matching algorithm.55
Ethics approval
This study was approved by the research ethics boards of St. Michael’s Hospital (no. 19-113), Toronto, and University of Toronto (no. 00038757).
Results
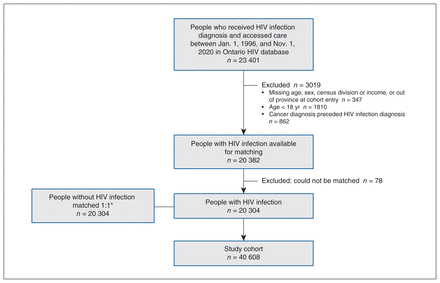
We matched 20 304 people infected with HIV to 20 304 people without HIV infection, for a total sample of 40 608 study participants (Figure 1). Most were males born in Canada or long-term residents who lived in urban centres and in the lowest neighbourhood income quintile (Table 1). The median age at cohort entry was 37 (IQR 30–45) years. Compared to people without HIV infection at cohort entry, those with HIV infection were more likely to have a higher comorbidity burden and high expected resource use (Table 1).
Flow chart showing participant selection. *Matched on birth year (within 1 yr), sex, census division (rurality), neighbourhood income quintile and region of birth (Canadian born or immigrated to Canada before 1986, born outside Canada in a country with low HIV infection prevalence, or born outside Canada in a region with high HIV infection prevalence).
Characteristics of people from Ontario, Canada, with and without HIV infection at cohort entry, 1996–2020
There were 2437 incident first primary cancers over 449 975 person-years of observation (median 10 [IQR 5–17] yr from cohort entry), of which 1534 (62.9%) were diagnosed among people with HIV infection and 903 (37.0%) among those without HIV infection (see Appendix 1, Supplementary Table S2 for site-specific cancers). Among people with a diagnosis of cancer, those with HIV infection had a greater proportion of ADC and infection-related NADC diagnoses (e.g., anal cancer) and a lower proportion of infection-unrelated cancers than people without HIV infection (Table 2).
Distribution of incident first primary cancers diagnosed among people with and without HIV infection
Trends in incidence over time
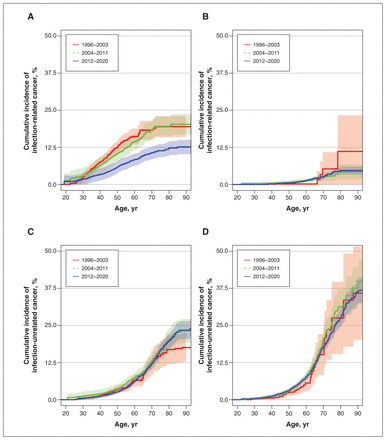
The incidence of infection-related cancer among people with HIV infection declined between the early (1996–2003) and the contemporary (2012–2020) cART eras (Figure 2A), with the cumulative risk of being diagnosed with an infection-related cancer by age 65 years declining from 19.0% (95% CI 15.6%–22.3%) to 10.0% (95% CI 7.9%–12.1%) between these periods (Table 3) (see Appendix 1, Supplementary Table S3 for the cumulative risk by age 75 yr). This decrease was driven largely by a decline in ADC incidence, with a smaller decrease in the incidence of infection-related NADCs (Table 3). We saw no change in trends in infection-related cancer among people without HIV infection (Figure 2B). We also saw no change in trends for infection-unrelated cancers among people with (Figure 2C) and without (Figure 2D) HIV infection.
Cumulative incidence of infection-related cancer and infection-unrelated cancer among people with (A, C) and without (B, D) HIV infection, by calendar period and age.
Cumulative risk of being diagnosed with an infection-related cancer by age 65 years for people with and without HIV infection, by calendar period
Among people with and without HIV infection, we observed no trends over time for prostate, lung or colorectal cancer (Appendix 1, Supplementary Figure S1). However, the risk of a diagnosis of anal or liver cancer by age 65 years and by age 75 years was higher among people with HIV infection than HIV-negative people, and this was consistent over time (Table 3; Appendix 1, Supplementary Table S3).
Age-specific trends
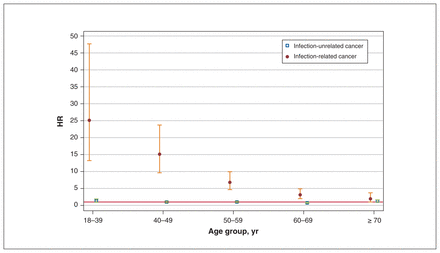
Averaged across all ages and time periods, the HR of infection-related cancer at any age for HIV-infected people was 7.9 (95% CI 6.2–10.1) times higher than that for people without HIV infection. However, the magnitude of the heightened risk decreased with increased age: the HR of infection-related cancer was 25.1 (95% CI 13.2–47.7) for those aged 18–39 years and 1.9 (95% CI 1.0–3.7) for those aged 70 years or older (Table 4 and Figure 3). The E-value we calculated, 15.3, would completely explain away an HR of 7.9, which suggests that the confounder needed would have to be 15.3 times more likely to occur in people with than without HIV infection and increase the risk of cancer by a factor of 15.3. E-values of 49.7 and 3.2 were calculated for HRs of 25.1 and 1.9, respectively. We found comparable trends in infection-related cancer in analyses stratified by calendar period (Appendix 1, Supplementary Table S4). The cause-specific HRs of infection-unrelated cancer were similar for people with and without HIV infection, with no clear and consistent trends by age or sex (Table 4).
Cause-specific hazard ratios for infection-related and infection-unrelated cancers comparing people with and without HIV infection, by age group and sex*
Cause-specific hazard ratios (HRs) of infection-unrelated and infection-related cancer for people with and without HIV infection, by age group. Associated 95% confidence intervals (CIs) are represented by whiskers. Red line represents null value of HR = 1.
Sex-specific trends
Relative to females without HIV infection, females with HIV infection had an HR of infection-related cancers; the relative hazard was significantly greater for those aged 18–39 years and 50–59 years (Table 4). Males with HIV infection had a higher HR of infection-related cancers than males without HIV infection, although the magnitude decreased with increasing age.
Males with HIV infection had a decreased cumulative risk of infection-related cancer in 2012–2020 compared to 1996–2003 and 2004–2011; this trend was not observed among females with HIV infection (Table 3; Appendix 1, Supplementary Table S3 and Supplementary Figure S2). Compared to females and males without HIV infection, females and males with HIV infection had an increased lifetime risk of infection-related cancers (Table 3; Appendix 1, Supplementary Table S3 and Supplementary Figure S2). We observed no differences by sex or calendar period for infection-unrelated cancers.
Interpretation
In our matched population-based study of people with and without HIV infection in Ontario from 1996 to 2020, we observed that the hazard of infection-related cancer was 7.9 times higher among people with HIV infection than among those without HIV infection. However, for people with HIV infection, the risk of having a diagnosis of infection-related cancer by age 65 decreased from 19% in the early cART era (1996–2011) to 10% in the contemporary cART era (2012–2020), a decline that was driven primarily by a decrease in ADCs. We attribute this finding to improvements in antiretroviral therapies over time and earlier commencement of HIV care, strategies that improve immune function and decrease the risk of persistent oncogenic infections.7 Nevertheless, we observed a consistently heightened risk of infection-related NADCs, particularly anal and liver cancers, among people with HIV infection. Vaccination against pathogens that cause such cancers, notably HPV and hepatitis B virus, is recommended for HIV-infected people.56 Moreover, there is strong evidence that anal cancer can be prevented through screening for and subsequent treatment of high-grade squamous intraepithelial lesions, which highlights the need for optimized screening and treatment of cancer precursors.57,58 Likewise, prevention of liver cancer includes treatment of hepatitis B virus and hepatitis C virus infection, as well as efforts tackling metabolic risk factors associated with nonalcoholic fatty liver disease–related liver cancer (e.g., diabetes, hypertension, cardiovascular disease).59 Consistent with other studies,20,60 we did not observe differences by HIV status in the risk of infection-unrelated cancers.
When we examined age-specific trends, we found that, relative to people without HIV infection, HIV-infected people had a greater hazard of infection-related cancer, which was more pronounced at younger ages. This is in keeping with earlier studies showing an earlier age at cancer diagnosis for certain cancers, including anal, lung, prostate and oropharyngeal, and myeloma, among people with compared to those without HIV infection.61–63 However, the higher relative difference at younger ages compared to older ages is partially due to the fact that the incidence in the background population increases with increasing age.64 We noted no major differences when we examined trends in infection-related and infection-unrelated cancer by sex.
Well-designed studies are needed that directly compare the risk of cancer at specific sites between people with and without HIV infection, adjusting for important confounders and cancer risk factors, while also exploring subgroups such as age and sex, and examining racial disparities.
Limitations
Strengths of our study include use of a population-based matched cohort of infected and uninfected people in a setting with publicly funded health care. However, people with undiagnosed HIV infection (estimated to be about 11% of those with HIV infection)65 and people who did not access care would have been excluded from our analyses. For people who were lost to follow-up in administrative databases, our assumptions regarding the end of follow-up may have resulted in overestimation of person-time at risk and, thus, an underestimate of cancer incidence. The resource use variable included resources used by people with HIV infection, which may have attenuated the effect estimates. Having dedicated funding support for community engagement in the study facilitated integrated engagement and contributed to the positive impact of patient and public engagement initiatives.
Given our large sample and long follow-up duration, we were able to capture many events and examine sex- and age-specific trends over time. Nevertheless, even with many cancer events, we lacked precision to estimate cancer risks for certain age and sex groups, as well as for specific cancer sites beyond the most common ones. Owing to data constrains, we were unable to examine histologic study findings, which may have led to misclassification of some infection-related cancers. Our competing risk approach using age as the time scale to obtain a measure of lifetime cancer risk66 and reporting cumulative cancer risk by age 65 years and 75 years has both clinical and public health utility. Although we calculated age-specific cancer risk stratified by calendar period, it is possible that there were changes in cancer risk we were unable to capture because the cumulative incidence metric relied on the assumption that the age-specific risk of cancer was constant across birth cohorts.11 Finally, data pertaining to ethnic background and known cancer risk factors, including lifestyle factors (e.g., smoking, alcohol), family history and clinical data (e.g., CD4 status, coinfection with hepatitis B or C, antiretroviral therapy, viral load), were unavailable. However, in our sensitivity analysis, we explored the magnitude of confounding bias that would explain away the observed association between HIV status and infection-related cancer and found that an extremely strong confounder would be required, one that is a very strong risk factor for cancer and strongly associated with HIV status. Although we cannot completely rule out confounding by such a risk factor, it is improbable that the true association is null since a confounder that has an association of such magnitude with the exposure and the outcome is extremely unlikely.
Conclusion
Despite considerable improvements over time, people with HIV infection in Ontario remained at much greater risk for infection-related cancers than people without HIV infection across all age groups. Our findings emphasize the importance of promoting and encouraging early routine cancer prevention strategies, such as early linkage to HIV care and treatment, screening for and treatment of viral hepatitis, HPV vaccination, and screening for HPV-related anal and cervical cancers for HIV-infected people. Although the risk of infection-unrelated cancers was similar for people with and without HIV infection, infection-unrelated cancers contributed greatly to the overall cancer burden for HIV-infected people, especially at older ages. Clinicians are encouraged to have ongoing conversations about lifetime cancer risk with patients in HIV care, and encourage risk reduction strategies such as smoking cessation, and screening for breast and colorectal cancer.
Acknowledgements
The authors thank members of the community engagement committee, including Christian Hui, as well as their community partner agencies. They also thank Karl Everett at ICES Central for his contributions to data preparation.
Footnotes
Competing interests: Marek Smieja reports funding from the Canadian Institutes of Health Research, Air Canada and the Greater Toronto Airports Authority. No other competing interests were declared.
This article has been peer reviewed.
Contributors: Ann Burchell (principal investigator) and Ioana Nicolau were involved in the study concept and design. Ioana Nicolau analyzed the data with supervision from Ann Burchell, Tony Antoniou, Jennifer Brooks and Rahim Moineddin. Ioana Nicolau drafted the manuscript. All authors interpreted the data, revised the manuscript critically for important intellectual content, approved the final version to be published and agreed to be accountable for all aspects of the work.
Funding: This study was funded by an Ontario HIV Treatment Network (OHTN) Endgame Game Changer grant (EFP-1104-GC). Ioana Nicolau was funded by a Canadian Institutes of Health Research (CIHR) Doctoral Award (201710MDR) and the Canadian HIV Observational Cohort Scholar Award (711352). Ann Burchell is a Canada Research Chair in Sexually Transmitted Infection Prevention (Tier 2). During the study, she received support from an OHTN Endgame Leader Award and a Non-Clinician Researcher Award from the Department of Family and Community Medicine, Faculty of Medicine, University of Toronto. Jennifer Gillis received support from the OHTN Endgame Student Leadership Award. Claire Kendall was supported by a CIHR New Investigator Award and a Clinical Research Chair Award from the Faculty of Medicine, University of Ottawa.
Data sharing: The data set from this study is held securely in coded form at ICES. Although legal data-sharing agreements between ICES and data providers (e.g., health care organizations and government) prohibit ICES from making the data set publicly available, access may be granted to those who meet prespecified criteria for confidential access, available at https://www.ices.on.ca/DAS (email: das{at}ices.on.ca). The full data set creation plan and underlying analytic code are available from the authors on request, with the understanding that the computer programs may rely on coding templates or macros that are unique to ICES and are therefore either inaccessible or may require modification.
Supplemental information: For reviewer comments and the original submission of this manuscript, please see www.cmajopen.ca/content/11/5/E894/suppl/DC1.
Disclaimer: Parts of this material are based on data and/or information compiled and provided by the Canadian Institute for Health Information; Immigration, Refugees and Citizenship Canada current to March 2020; and Ontario Health (Cancer Care Ontario). This study was supported by ICES, which is funded by an annual grant from the Ontario Ministry of Health (MOH) and the Ministry of Long-Term Care (MLTC). The analyses, results, conclusions, opinion, views and statements expressed herein are solely those of the authors and do not reflect those of the funding or data sources; no endorsement is intended or should be inferred.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/
References
- © 2023 CMA Impact Inc. or its licensors