Abstract
Background: Canadian immunization programs for rotavirus started in 2011. We sought to determine their effect on the burden of community-acquired admissions and hospital-acquired rotavirus at pediatric hospitals.
Methods: The Canadian Immunization Monitoring Program Active (IMPACT) network conducted active surveillance for rotavirus-positive hospital admissions between 2005 and 2020 at 12 pediatric hospitals. We used yearly rates of community-acquired rotavirus per 10 000 admissions and hospital-acquired rotavirus infections per 1000 patient-days to determine changes in the pre- and post-vaccine program periods.
Results: During the 15-year study period, 5691 rotavirus hospital admissions and hospital-acquired infections were detected, including 4323 (76%) community-acquired infections and 1368 (24%) hospital-acquired infections. The average community-acquired rate in the pre-vaccine period was 60.3 (95% confidence interval [CI] 53.7–68.3) per 10 000 admissions, with a decline to 11.0 (95% CI 7.5–15.1) per 10 000 admissions in the post-vaccine period, resulting in an average reduction of 81.7% (95% CI 74.4%–87.8%). The rate of hospital-acquired rotavirus declined from 0.35 (95% CI 0.29–0.41) per 1000 patient-days in the pre-vaccine period to 0.05 (95% CI 0.03–0.07) per 1000 patient-days in the post-vaccine period, resulting in an 85.3% (95% CI 77.7%–91.9%) average decline. Herd protection was present among children aged 2–16 years.
Interpretation: Although start dates of rotavirus vaccine programs across provinces varied, there was around an 80% average decrease in both community-acquired and hospital-acquired rotavirus infections at pediatric hospitals in Canada in the 1- to 9-year interval after implementation of rotavirus vaccine programs. Herd protection is an important aspect of rotavirus vaccines for other children who are not vaccine eligible, and rotavirus vaccines continue to provide important benefits both for children and health care systems.
Rotavirus is a major cause of pediatric gastroenteritis worldwide with severe manifestations in 1%–2% of infected infants.1 Before the availability of the rotavirus vaccine, it was estimated that there were between 4500 and 10 000 hospital admissions for rotavirus annually in Canada and an economic analysis concluded that immunization would be cost-effective.2,3 Two live attenuated vaccines, (monovalent Rotarix [i.e., Rot-1] from GlaxoSmithKline Biologicals, given at 2 and 4 mo of age, and pentavalent RotaTeq [i.e., Rot-5] from Merck, given at 2, 4 and 6 mo of age) are approved for infants in Canada. Provincial and territorial publicly funded rotavirus vaccine programs started in 2011. The Canadian national immunization coverage report in 2021 reported 85.6% coverage for 2 doses of rotavirus vaccine, while provinces have reported 72%–84.2% coverage.4–8
Rotavirus vaccine programs were introduced in United States, Australia and the United Kingdom in 2006, 2007 and 2013, respectively, resulting in decreases in rates of hospital admission of 75%–80%.9–13 A Canadian study using administrative discharge data from 7 provinces determined that, for children younger than 3 years, hospital admissions for rotavirus gastroenteritis had decreased 53%–75% within 2 years of starting a program.14 The aim of this study was to determine the impact of publicly funded rotavirus immunization programs on both admissions for community-acquired rotavirus infections and hospital-acquired rotavirus at tertiary care pediatric hospitals in Canada. We also sought to determine vaccine failures and estimates of herd protection in the post-vaccine period.
Methods
Setting
Active surveillance for hospital admissions for community-acquired rotavirus infections and hospital-acquired rotavirus infections was conducted by 12 pediatric hospitals from 2005 to 2020 through the Canadian Immunization Monitoring Program, Active (IMPACT) surveillance network, which covers 50% of pediatric hospital admissions in Canada and 90% of tertiary pediatric care beds.15 The Rot-1 vaccine (Rotarix) was used primarily until mid-2018, after which Rot-5 (Rotateq) was used through 2020. The month and year of initiation of publicly funded immunization programs in Canada, including provinces and territories without IMPACT sites, is shown in Table 1. The IMPACT program collects total hospital admissions and hospital inpatient days (excluding newborn nursery and mental health admissions) yearly.
Dates of introduction for publicly funded rotavirus immunization programs in Canada by province with and without IMPACT sites
Study participants
We included children aged 0–16 years who had laboratory-confirmed rotavirus infections who were admitted to an IMPACT hospital between Jan. 1, 2005, and Dec. 31, 2020. Since some sites restrict admission to patients 16 years or younger, we excluded older participants for all sites to avoid underrepresentation of patients aged 17 years or older.
Study design
Trained nurse monitors prospectively identified and listed any patients with gastroenteritis from admission and scanned microbiology reports for rotavirus on a regular basis. To ensure completeness of case ascertainment, audits of discharge codes from the International Classification of Diseases and Related Health Problems, 10th Revision were conducted annually to identify any missed cases using codes A08 (viral and other intestinal infections), A09 (diarrhea and gastroenteritis of infectious origin), K52.9 (noninfectious gastroenteritis), R11 (nausea and vomiting) and R15 (fecal incontinence). If found, these cases were screened for rotavirus positivity. We included patients if they had acute gastrointestinal symptoms (diarrhea with or without vomiting). We excluded patients with rotavirus-positive specimens detected less than 72 hours after admission who were admitted for unrelated reasons (e.g., trauma), as they were not admitted for gastroenteritis.
All centres used the same strategies and definitions for finding patients from 2005 onward. The case report form was also the same, except for the clinical variables that were modified as of 2008. Rotavirus from stool specimens was detected using either enzyme-linked immunoassay or molecular diagnostics (i.e., polymerase chain reaction).
We defined patients with laboratory confirmation of rotavirus less than 72 hours after hospital admission as having community-acquired infections. We defined rotavirus infections identified from patients 72 hours or more after admission or from those readmitted with rotavirus within 48 hours after the initial discharge as being hospital-acquired infections.
Data were abstracted from medical charts after patient discharge using a structured case report form. Data included demographics, pre-existing comorbid or immune-compromising conditions, clinical features (e.g., presence of dehydration, electrolyte abnormalities, seizures), length of stay, number and type of rotavirus vaccine received and outcome. Prematurity was defined as a child born at 36 weeks’ gestation or less who was still in the first year of life. The determination of dehydration was based on admission notes. If not recorded in the hospital chart, data were reported as not available.
We defined a vaccine failure as when a child with rotavirus gastroenteritis had received 2 doses of Rot-1 or 3 doses of Rot-5 vaccine at least 2 weeks before admission.
Statistical analysis
We reported the overall, age-specific and site-specific rates of community-acquired rotavirus admission per 10 000 hospital admissions. We calculated the rates of hospital-acquired rotavirus infections per 1000 inpatient days. We stratified patients by age group (0–11 mo, 12–23 mo, 24–59 mo, 5–9 yr and 10–16 yr).
For each province, we designated the year rotavirus vaccine programs were introduced as year 0, with the years before vaccine programs being designated −15 to −1 and years following introduction designated as +1 to +9. We conducted all calculations with the year of vaccination program start as year 0, including sites where the program started in November or later where the next calendar year was designated as year 0, as this would allow time for program roll-out. We excluded patients with rotavirus in year 0 cases in the pre- and post-vaccine periods calculations. To calculate rate reduction, we determined the average rates for the pre- and post-vaccine eras. Each year contributed 1 data point. We used the change between these 2 rates to determine the percentage for the rate reduction. We estimated confidence intervals (CIs) for the average rates and percentage changes between rates using a nonparametric bootstrap with 100 000 replications.
Analysis for indirect effect of the vaccine included only individuals within age groups who were previously or currently ineligible for vaccine based on age.16 We calculated average rates of rotavirus hospital admissions for community-acquired rotavirus infections and hospital-acquired rotavirus infections in these eligible cohorts separately for the pre- and post-vaccine periods.
We calculated proportions of patients with clinical features and ICU admissions, and average lengths of stay for each of the pre- and post-vaccine eras among patients with community-acquired rotavirus only, since patients with hospital-acquired infections may have confounding symptoms related to pre-existing conditions or treatments.
We conducted analyses in R (version 4.1.1; The R Project for Statistical Computing) and data cleaning in SAS (version 9.4, SAS Institute).
Ethics approval
We obtained research or hospital ethics board approval at all sites.
Results
A total of 5691 rotavirus hospital admissions and hospital-acquired infections were identified at the 12 sites from Jan. 1, 2005, to Dec. 31, 2020, of which 4323 (75.9%) were community-acquired infections and 1368 (24.0%) were hospital-acquired infections. Overall, the median age for community-acquired infections was 1.5 (interquartile range [IQR] 0.8–2.9) years and 0.8 (IQR 0.3–2.0) years for hospital-acquired infections. Of the 5691 infections, 2148 (37.7%) were among children aged 0–11 months, 1523 (26.7%) were among those aged 12–23 months, 1305 (22.9%) were among those aged 24–59 months, 479 (8.4%), were among those aged 5–9 years and 236 (4.1%) were among children aged 10–16 years. Of the entire cohort of 5691 children, 2621 (46.0%) had underlying comorbidities (Appendix 1, available at www.cmajopen.ca/content/11/6/E1156/suppl/DC1) and among this group, 790 (13.9%) were immunosuppressed.
Comparisons of pre- and post-vaccine periods
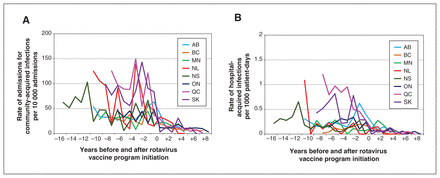
The rates between periods were based on a comparison of the mean of the 15 pre-implementation rates and the mean of the 9 post-implementation rates. The overall average rate of community-acquired rotavirus admissions decreased from 60.3 (95% CI 53.7–68.3) per 10 000 admissions in the pre-vaccine period to 11.0 (95% CI 7.5–15.1) per 10 000 admissions in the post-vaccine period, resulting in a rate reduction of 81.7% (95% CI 74.4%–87.8%). The prevalence of hospital-acquired rotavirus per 1000 patient-days decreased by 85.3% (95% CI 77.7%–91.9%) from 0.35 (95% CI 0.29–0.41) in the pre-vaccine period to 0.05 (95% CI 0.03–0.07) per 1000 patient-days in the post-vaccine period. Given the large observed decrease in rates, an analysis based on actual counts that also accounted for interprovincial differences and autocorrelation in rates may have resulted in CIs that are different from the ones presented here, but the magnitude would be similar. The average rates by province in the pre- and post-vaccine periods are illustrated in Figure 1.
(A) Average yearly rate of community-acquired rotavirus admissions per 10 000 hospital admissions and (B) average yearly rate of hospital-acquired rotavirus infections per 1000 patient-days by province from 2005 to 2020, in the pre- and post-vaccine periods, excluding year of program implementation (year 0).
The demographics of patients with community- and hospital-acquired rotavirus by pre- and post-vaccine period are seen in Table 2. The overall proportion of community-acquired patients aged 24 months or older in the pre-vaccine period was 35%, compared with 56% in the post-vaccine period. Among 3331 patients with community-acquired infections in the pre-vaccine period, 2277 (68.4%, 95% CI 66.8%–69.0%) were generally healthy, versus 372 (53.4%, 95% CI 52.9%–60.4%) of 656 in the post-vaccine period. Among 1109 patients with hospital-acquired infections, in the pre-vaccine period, 190 (17.1%, 95% CI 15.0%–19.5%) were healthy, compared with 9 (5.5%, 95% CI 3.0%–10.2%) of 162 in the post-vaccine period. Comparison of clinical features in patients with community-acquired disease in pre- and post-vaccine periods is seen in Table 3.
Ages of patients with community-acquired rotavirus infections admitted to hospital or hospital-acquired rotavirus infection in the pre-vaccine versus post-vaccine time periods*
Comparison of clinical presentations and hospital admission for patients with community-acquired rotavirus infections (2008–2020) in the pre-vaccine and post-vaccine periods, excluding those from year of vaccine implementation (year 0)
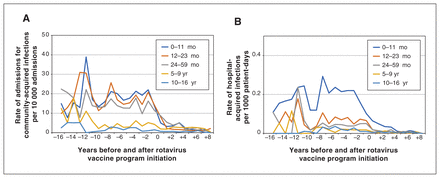
The yearly average rates in the pre- and post-vaccine periods by age is illustrated in Figure 2. Among vaccine-ineligible children, the average admission rates for community-acquired infections decreased 74%–91%, and the rate of hospital-acquired infections decreased 61%–95% (Table 4).
(A) Average yearly rate of community-acquired rotavirus admissions per 10 000 hospital admissions and (B) average yearly rate of hospital-acquired rotavirus infections per 1000 patient-days by age group from 2005 to 2020, in the pre- and post-vaccine periods, excluding year of program implementation (year 0).
Average rates of hospital admission for community-acquired rotavirus infection and hospital-acquired rotavirus infection among age groups that were not eligible for the vaccine in the pre- and post-vaccine periods
In the post-vaccine period, 61 (7.4%) of 818 patients met the definition of vaccine failure; of these, 53 (86.9%) were community-acquired infections and 8 (13.1%) were hospital-acquired infections. The mean age for patients with vaccine failures was 1.9 (standard deviation [SD] 1.5) years and the median was 1.7 (IQR 0.9–2.4) years. Of these 61 patients, 27 (44.2%) had underlying comorbidities, of which 9 (33.3%) had immunocompromising conditions.
During the entire surveillance period, a total of 40 (0.7%) deaths, including 13 (32.5%) community-acquired infections and 27 (67.5%) hospital-acquired infections, were reported during a hospital admission. During the pre-vaccine period, the average mortality rate was 0.25 per 10 000 admissions and was 0.18 per 10 000 admissions during the post-vaccine period. Of these 40 deaths, 39 (97.5%) had underlying diseases or prematurity (one was unknown) with 26 (65.0%) occurring in the pre-vaccine period (−1 to −15 yr), 4 (10%) occurring the year of program start (yr 0) and 10 (25%) occurring in the post-vaccine program period (+1 to +9 yr). Of the 10 deaths in the post-vaccine period, 6 were community-acquired infections (3 patients aged 0–23 mo, 3 patients aged 24–59 mo) and 4 were hospital-acquired infections (all patients aged 0–11 mo); 7 had had no rotavirus vaccines, 2 had received 1 dose and 1 had received the complete series. It is unknown if rotavirus infection contributed to the deaths.
Interpretation
This 15-year, real-world, active surveillance for rotavirus infections in 12 Canadian pediatric hospitals demonstrates a substantial and sustained decrease of 81.7% for community-acquired rotavirus admissions and 85.3% for hospital-acquired rotavirus infections among children up to 16 years of age since rotavirus vaccines were introduced into provincial vaccine programs in 2011. Children aged 2–16 years who were not eligible to receive the vaccine showed a 74%–91% decrease in admission rates for community-acquired rotavirus and a 61%–95% decrease in hospital-acquired infections. The vaccine failure rate in this cohort was 7.4%.
Data from low-mortality countries have shown a vaccine efficacy of 90% against admissions for severe disease among children younger than 3 years, especially in the first years of programs, but did not include Canadian data.17,18 Administrative and hospital data from both Ontario and Quebec 1.5–5 years after implementation of vaccine programs also showed declines of 53%–90% in hospital admissions for rotavirus among children aged 2–5 years,14,19,20 supporting a comparable long-term reduction rate of just over 80% for hospital admissions. These data are comparable to reports from Finland, England, Austria, the US, New Zealand and Australia documenting decreases of 75%–92% in rotavirus admissions, mainly in the first 5–6 years of program implementation.21–26 Data from 10–15 years after the availability of the rotavirus vaccine in the US continued to show an 80% drop in admissions despite estimates of rotavirus vaccine coverage of 73.2%.27,28 Similar to other reports, we saw an increase in the proportion of children older than 2 years admitted to hospital for community-acquired disease in the post-vaccine period, compared with the pre-vaccine period.12,29 In New York, in the 9-year post-vaccine period 15% of hospital admissions were among children older than 5 years, compared with only 3.7% in the pre-vaccine period.30 Given high vaccine rates in Canada, and as more cohorts receive the vaccine, it is expected that these age differences will disappear if there is continued high vaccine uptake and no substantial genotype shifts that may jeopardize efficacy.31
Hospital-acquired rotavirus infections often affect the most vulnerable children in hospital and has substantial cost implications for pediatric hospitals through increased length of stay.32 A previous IMPACT study had shown that 19%–27% of rotavirus infections in hospitals were acquired in hospital.33 This study and others highlight substantial declines in hospital-acquired rates very rapidly after program implementation, especially among children aged 0–11-months, suggesting that community-acquired rotavirus admissions are likely a major source of transmission to admitted patients.34–36
Herd immunity refers to a reduction in disease among unvaccinated, susceptible people due to decrease in disease among vaccinated people, which is influenced by exposure to other infected children or previous immunity.16,37 We saw substantial reductions in hospital admissions for community-acquired rotavirus and hospital-acquired rotavirus among non–vaccine-eligible age groups, similar to studies 3–4 years after the introduction of rotavirus vaccines, which noted substantial indirect protection of 82%–90% among young children38,39 In Australia and the US, including a study 7 years after the rotavirus programs started, rotavirus admissions decreased in all age groups, including adults, supporting substantial herd protection, likely due to decreased transmission from infants who have been vaccinated.40–43 A study in Finland, however, reported rotavirus disease in nonvaccine-eligible cohorts 4–6 years after rotavirus vaccine programs, especially outside epidemic seasons, despite high vaccine rates.44
Knowledge gaps include whether children vaccinated in infancy will develop more or less severe disease later in life compared with cohorts with wild-type infections. As vaccinated cohorts increase in size over time, dynamics of rotavirus disease transmission are unknown. It is also not known whether rotavirus genotypes will shift over time, with potential vaccine breakthrough, and whether new types of vaccines will be needed to prevent this hitherto common childhood disease. Genotype analysis is currently ongoing.
Limitations
Limitations include under-representation from provinces and areas that do not have IMPACT surveillance programs and from community hospitals with a change in admission patterns over time. Rates of rotavirus infection vary as case mix and size of Canadian pediatric hospitals are different by region, but the sites in this study did not change over time. It is also possible that some patients did not have samples sent for laboratory analysis, thereby underestimating the total number of infections. Rotavirus infections from children admitted from other hospitals could have been acquired in hospital rather than in the community, thereby underestimating hospital-acquired infections or overestimating community-acquired infections.
Conclusion
The publicly funded rotavirus immunization programs in Canada have contributed to decreases in community- and hospital-acquired rotavirus infections in pediatric hospitals of more than 80%, including similar reductions in disease among admitted children in pediatric hospitals as well as herd protection in children aged 0–16 years, over 9 years since program implementation. As such, the rotavirus vaccine provides substantial health benefits for children in reducing morbidity and are likely cost saving for children’s hospitals.
Footnotes
Members of Immunization Monitoring Program, Active: Karina A. Top MD, Dalhousie University and Canadian Center for Vaccinology, IWK Health Centre, Halifax, NS; Roseline Thibeault MD, Centre Mère-Enfant Soleil de Québec (Pavillon CHUL), Université Laval, Québec City, Que.; Shaun K. Morris MD, Hospital for Sick Children, Dalla Lana School of Public Health, University of Toronto; Kescha Kazmi MD, Hospital for Sick Children, Temerty Faculty of Medicine, University of Toronto, Toronto, Ont.; J. Bullard MD, Winnipeg Children’s Hospital Health Sciences Centre, University of Manitoba, Winnipeg, Man.; Laura Sauvé MD, BC Children’s Hospital, University of British Columbia, Vancouver, BC; C. Constantinescu MD, Alberta Children’s Hospital, Pediatric Infectious Disease, University of Calgary, Calgary, Alta.; Jesse Papenburg MD, Montreal Children’s Hospital, McGill University; Marc Lebel MD, CHU-Sainte-Justine, Montréal, Université de Montréal; Fatima Kakkar MD, CHU-Sainte-Justine, Centre de recherche du CHU Sainte-Justine, Montréal, Que.; Cheryl Foo MD, Janeway Children’s Health and Rehabilitation Center, Memorial University, St. John’s, NL; Catherine Burton MD, Stollery Children’s Hospital and University of Alberta, Edmonton, Alta.; Rupeena Purewal and Rupesh Chawla, Jim Pattison Children’s Hospital, University of Saskatchewan, Saskatoon, Sask.
Competing interests: Julie Bettinger reports research funding from the Canadian Institutes of Health Research (CIHR), the British Columbia Ministry of Health, the Public Health Agency of Canada (PHAC) and the COVID-19 Immunity Task Force; travel support from the Public Health Agency of Canada and the Canadian Public Health Association; and volunteer roles with the National Advisory Committee on Immunization and the BC Immunization Committee. Manish Sadarangani reports research funding from GSK, Merck, Moderna, Pfizer, Sanofi-Pasteur and Symvivo. He is chair or deputy chair of 2 data safety monitoring boards for SARS-CoV-2 vaccine trials, involving different vaccines. Scott Halperin reports research funding from GSK, Merck, Sanofi, Moderna, Entos, the Vaccine and Infectious Disease Organization, CIHR, the National Institutes of Health, the United States Centers for Disease Control and Prevention, Pfizer, PHAC, CanSino and Inventprise; consulting fees for serving on ad hoc advisory boards for Merck, Sanofi, Pfizer, AstraZeneca, NovaVax and GSK; payment for expert testimony from the Canadian Medical Protective Association and the Province of Ontario; and participation on data safety monitoring boards for the International AIDS Vaccine Initiative and Medicago. No other competing interests were declared.
This article has been peer reviewed.
Contributors: Nicole Le Saux, Julie Bettinger and Scott Halperin contributed to the conception and design of the work. Nicole Le Saux, Julie Bettinger, Manish Sadarangani, Taj Jadavji and Scott Halperin contributed to data acquisition. Julie Bettinger, Hennady Shulha, Doug Coyle and Timothy Booth contributed to data analysis. Nicole Le Saux, Julie Bettinger, Manish Sadarangani, Doug Coyle and Scott Halperin contributed to data interpretation. Nicole Le Saux drafted the manuscript. All of the authors revised it critically for important intellectual content, gave final approval of the version to be published and agreed to be accountable for all aspects of the work.
Funding: The Canadian Immunization Monitoring Program Active (IMPACT) network of pediatric investigators is managed by the Canadian Paediatric Society, which receives ongoing funding from the Public Health Agency of Canada’s Centre for Immunization and Respiratory Infectious Diseases for IMPACT, a national surveillance initiative. Funding for this specific surveillance project was provided by the Public Health Agency of Canada, the Canadian Paediatric Society and through unrestricted grants from GSK Pharmaceuticals and Merck Canada.
Data sharing: Data used for this study cannot be shared.
Supplemental information: For reviewer comments and the original submission of this manuscript, please see www.cmajopen.ca/content/11/6/E1156/suppl/DC1.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/
References
- © 2023 CMA Impact Inc. or its licensors