Abstract
Background: Hypertensive disorders, especially preeclampsia, are the leading reason for provider-initiated preterm birth. We estimated how universal acetylsalicylic acid (ASA) prophylaxis might reduce rates of provider-initiated preterm birth associated with preeclampsia and intrauterine growth restriction, which are related conditions.
Methods: We performed a cohort study of singleton hospital births in 2013 in Canada, excluding Quebec. We estimated the proportion of term births and provider-initiated preterm births affected by preeclampsia and/or intrauterine growth restriction, and the corresponding mean maternal and newborn hospital length of stay. We projected the potential number of cases reduced and corresponding hospital length of stay if ASA prophylaxis lowered cases of preeclampsia and intrauterine growth restriction by a relative risk reduction (RRR) of 10% (lowest) or 53% (highest), as suggested by randomized clinical trials.
Results: Of the 269 303 singleton live births and stillbirths in our cohort, 4495 (1.7%) were provider-initiated preterm births. Of the 4495, 1512 (33.6%) had a diagnosis of preeclampsia and/or intrauterine growth restriction. The mean maternal length of stay was 2.0 (95% confidence interval [CI] 2.0-2.0) days among term births unaffected by either condition and 7.3 (95% CI 6.1-8.6) days among provider-initiated preterm births with both conditions. The corresponding values for mean newborn length of stay were 1.9 (95% CI 1.8-1.9) days and 21.8 (95% CI 17.4-26.2) days. If ASA conferred a 53% RRR against preeclampsia and/or intrauterine growth restriction, 3365 maternal and 11 591 newborn days in hospital would be averted. If ASA conferred a 10% RRR, 635 maternal and 2187 newborn days in hospital would be averted.
Interpretation: A universal ASA prophylaxis strategy could substantially reduce the burden of long maternal and newborn hospital stays associated with provider-initiated preterm birth. However, until there is compelling evidence that administration of ASA to all, or most, pregnant women reduces the risk of preeclampsia and/or intrauterine growth restriction, clinicians should continue to follow current clinical practice guidelines.
Preterm birth, defined as birth before 37 weeks' gestation, occurs among 6%-11% of all viable pregnancies globally.1 In Canada, preterm birth rates are about 8%, with no evidence of decline.2 Preterm birth remains the leading cause of infant mortality, making its reduction and an increased understanding of its causes laudable goals.
Globally, an estimated 30%-35% of preterm births are provider-initiated - otherwise termed "iatrogenic" or "indicated" - with rates as high as 50% in many high-income countries.3,4 Provider-initiated preterm birth occurs through induction of labour or prelabour cesarean delivery, typically owing to a maternal and/or fetal indication, such as hypertensive disorders of pregnancy, intrauterine growth restriction or antepartum hemorrhage. Among these, hypertensive disorders, especially preeclampsia, are the leading reason for provider-initiated preterm birth.4,5 One approach to reducing rates of provider-initiated preterm birth, therefore, is to lower the incidence of preeclampsia and intrauterine growth restriction, which are related conditions.6,7
Clinical practice guidelines in Canada and elsewhere recommend treatment with daily low-dosage (60-162 mg) acetylsalicylic acid (ASA) for the prevention of preeclampsia and intrauterine growth restriction in women at increased risk for preeclampsia,8-10 based on convincing evidence from randomized clinical trials.11-13 ASA treatment initiated at 12 to 28 weeks' gestation and continued until delivery may reduce preterm preeclampsia rates by as much as 89% (95% confidence interval [CI] 67%-96%)14 and rates of intrauterine growth restriction by 56% (95% CI 35%-70%).12 Despite the evidence and recommendations, however, the level of adoption of ASA prophylaxis in Canada is likely low, as elsewhere.15,16 Thus, an opportunity to reduce rates of preeclampsia and fetal intrauterine growth restriction and, accordingly, provider-initiated preterm birth is not being realized.
Clinical prediction models for preeclampsia17 and intrauterine growth restriction18 typically generate discriminative estimates of only 0.75, which are suboptimal in identifying pregnancies at high risk. In addition, over 50% of cases of preterm preeclampsia may not be preceded by any known risk factor(s).19 ASA is extremely inexpensive, and its adverse effects for the woman and the fetus appear negligible.20,21 Moreover, although not currently recommended, ASA prophylaxis could be considered for all pregnant women who are not ASA intolerant, including those at low risk.22 Given the low rate of ASA hypersensitivity in the general population,23 we investigated the degree to which a universal ASA strategy might reduce rates of preeclampsia and intrauterine growth restriction and, thus, the number of provider-initiated preterm births. We further estimated how such reductions in provider-initiated preterm birth would affect maternal and newborn hospital length of stay.
Methods
Setting and design
We performed a retrospective population-based cohort study of all singleton live-born or stillborn hospital births in 9 Canadian provinces and the 3 territoriesin 2013. We obtained all information from the Canadian Institute for Health Information's Discharge Abstract Database, which captures administrative, clinical and demographic information on hospital discharges. Data were excluded for the province of Quebec, which does not report to the Discharge Abstract Database.
Maternal records included deliveries of live-born and stillborn infants. Newborn records were restricted to live births, as newborn length of stay was 1 of the study outcome measures. We deterministically linked maternal and newborn records based on the woman's hospital chart number, which is also recorded on the newborn's chart, and the woman's province of residence. A linkage rate of 97% was achieved. Analyses were restricted to girls and women aged 14-50 years and to births of infants of 24-42 weeks of gestational age weighing 500 g or more. Gestational age in the Discharge Abstract Database is derived from the best clinical estimate recorded in the medical chart, based on ultrasound dating or the last menstrual period.24
Exposure and outcome variables
We defined provider-initiated preterm birth as 1) birth between 24 and 36 completed weeks' gestation and 2) the absence of a diagnosis of preterm spontaneous labour with preterm delivery (International Statistical Classification of Diseases, 10th Revision [ICD-10] diagnostic code O601), preterm labour with term delivery (ICD-10 O602), premature rupture of membranes (ICD-10 O42) or delayed delivery after spontaneous or unspecified rupture of membranes (ICD-10 O756) and 3) the presence of a cesarean delivery (Canadian Classification of Health Interventions intervention code 5MD60) and/or induction of labour (Canadian Classification of Health Interventions 5AC30).
We defined preeclampsia as the presence of a maternal ICD-10 code O11, O14 or O15 at the index birth. We used sex-specific birth weight under the third percentile at a given gestational age as a proxy for intrauterine growth restriction;25,26 if birth weight was missing, we identified intrauterine growth restriction using ICD-10 diagnostic code O365 (maternal care for restricted fetal growth). We chose the cut-point of the third percentile as it represents a more pathological threshold at which mortality and morbidity are significantly increased.27
Statistical analysis
Our analyses were underpinned by the following assumptions: 1) prevented cases of provider-initiated preterm birth would advance to term birth without preeclampsia or intrauterine growth restriction, 2) ASA treatment homogeneously prevents preeclampsia or intrauterine growth restriction of any severity, 3) provider-initiated preterm birth accompanied by preeclampsia and/or intrauterine growth restriction is due to 1 or both of these conditions and 4) current rates of preeclampsia and intrauterine growth restriction reflect low use of ASA prophylaxis in pregnancy.
We estimated the proportion of term and provider-initiated preterm births affected by 4 mutually exclusive combinations of preeclampsia and intrauterine growth restriction: 1) neither preeclampsia nor intrauterine growth restriction, 2) preeclampsia and no intrauterine growth restriction, 3) intrauterine growth restriction and no preeclampsia or 4) both preeclampsia and intrauterine growth restriction. We then determined the mean maternal and newborn hospital length of stay in each of the 4 groups, for all provider-initiated preterm births at 24-36 weeks' gestation and for those at 24-31 weeks (early), 32-34 weeks (moderate) and 35-36 weeks (late).
Next, to generate unadjusted estimates of the total number of maternal and newborn cases and of hospital days that might be prevented if all pregnant women took ASA prophylactically, we juxtaposed low and high relative risk reductions (RRRs) for the efficacy of ASA. We estimated the range of provider-initiated preterm births that would be prevented if ASA prophylaxis conferred the lowest (10%)11 and highest (53%)12 RRR for preeclampsia and for small for gestational age or intrauterine growth restriction reported in meta-analyses of randomized controlled trials. We then multiplied the number of prevented cases of preeclampsia or intrauterine growth restriction by the mean number of hospital days for women and newborns following a term birth unaffected by preeclampsia or intrauterine growth restriction (the comparison group) to produce value "A," and multiplied the remaining number of cases by the mean number of hospital days for women and newborns with provider-initiated preterm birth affected by preeclampsia and/or intrauterine growth restriction (our cohort group) to produce value "B." The comparison group characterized our 4 assumptions. We calculated the annual net reduction in maternal and newborn hospital days by subtracting the sum of value A and value B from the total number of hospital days observed in our cohort.
We conducted analyses using SAS 9.1 (SAS Institute).
Ethics approval
No research ethics board submission was required for this project, as all analysis used denominalized hospital discharge abstract data.
Results
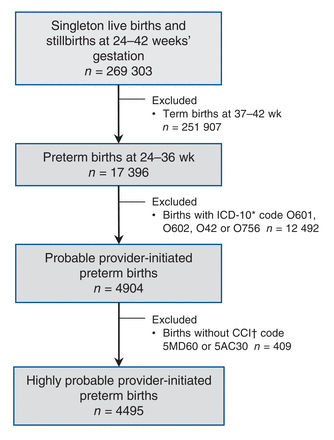
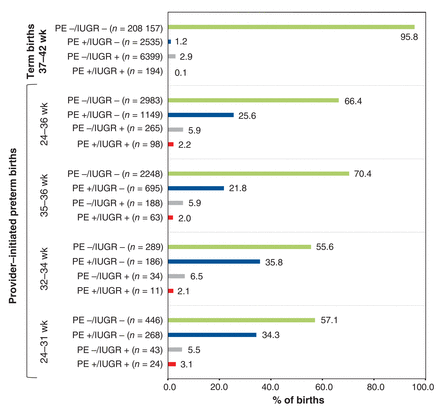
Of the 269 303 singleton live births and stillbirths in Canada (excluding Quebec) in 2013, 17 396 (6.5%) were preterm births, and 4495 (1.7%) were provider-initiated preterm birth - that is, 25.8% of all preterm births were provider-initiated preterm births (Figure 1). Of the 4495 provider-initiated preterm births, 1512 (33.6%) had a diagnosis of preeclampsia and/or intrauterine growth restriction; preeclampsia was much more prevalent than intrauterine growth restriction at any gestational age in this group (Figure 2).
Flow chart showing sample selection to create the cohort of 14- to 50-year-old girls/women with a singleton provider-initiated preterm live birth or stillbirth in 2013 in Canada, excluding Quebec. *International Statistical Classification of Diseases, 10th Revision (ICD-10) diagnostic code O601 = preterm spontaneous labour with preterm delivery, O602 = preterm labour with term delivery, O42 = premature rupture of membranes, O756 = delayed delivery after spontaneous or unspecified rupture of membranes. †Canadian Classification of Health Interventions (CCI) intervention code 5MD60 = Cesarean section delivery, 5AC30 = induction of labour.
Proportion of all term births and provider-initiated preterm births affected by preeclampsia/eclampsia (PE) and/or fetal intrauterine growth restriction (IUGR). Included are all 217 285 singleton term births, regardless of mode of delivery, and 4495 provider-initiated preterm births in Canada (excluding Quebec), 2013. Provider-initiated preterm births are classified as early (24-31 weeks), moderate (32-34 weeks) or late (35-36 weeks).
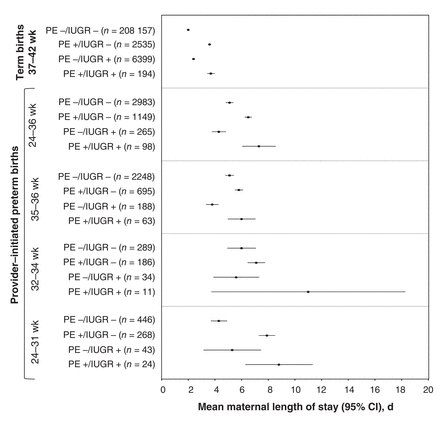
The mean maternal hospital length of stay was 2.0 (95% CI 2.0-2.0) days among term births unaffected by preeclampsia or intrauterine growth restriction, and 3.7 (95% CI 3.4-4.0) days among term births with both preeclampsia and intrauterine growth restriction (Figure 3). A similar, but more pronounced, pattern was seen among provider-initiated preterm births, with mean maternal length of stay increasing to 7.3 (95% CI 6.1-8.6) days in the presence of preeclampsia and intrauterine growth restriction (Figure 3).
Mean maternal hospital length of stay in the index delivery for all term and provider-initiated preterm births affected by preeclampsia/eclampsia (PE) and/or fetal intrauterine growth restriction (IUGR). Included are all 217 285 singleton term births, regardless of mode of delivery, and 4495 provider-initiated preterm births in Canada (excluding Quebec), 2013. Provider-initiated preterm births are classified as early (24-31 weeks), moderate (32-34 weeks) or late (35-36 weeks).
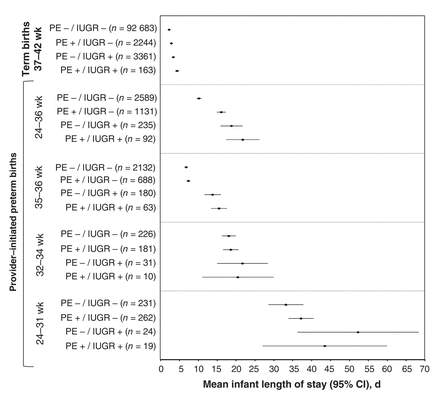
Compared with term newborns (mean length of stay 1.9 [95% CI 1.8-1.9] d), newborns with provider-initiated preterm birth had a much longer mean length of stay, especially those affected by both preeclampsia and intrauterine growth restriction (21.8 [95% CI 17.4-26.2] d) and those born more prematurely (Figure 4).
Mean newborn hospital length of stay during the index delivery for all term and provider-initiated preterm live births affected by preeclampsia/eclampsia (PE) and/or fetal intrauterine growth restriction (IUGR). Included are all 216 955 singleton term live births, regardless of mode of delivery, and 4047 provider-initiated preterm births in Canada (excluding Quebec), 2013. Provider-initiated preterm births are classified as early (24-31 weeks), moderate (32-34 weeks) or late (35-36 weeks).
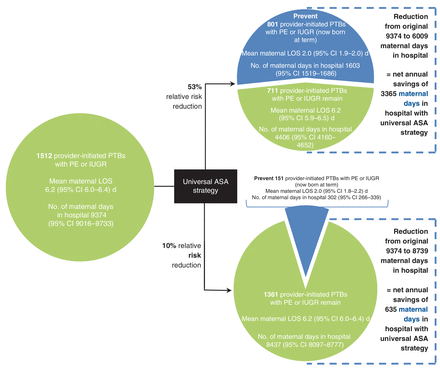
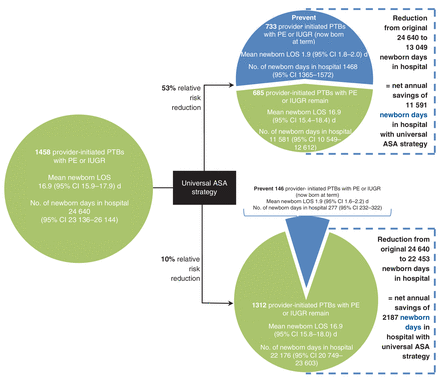
Under a universal ASA prophylaxis strategy, and assuming a 53% RRR conferred by ASA for preventing preeclampsia and/or intrauterine growth restriction, 801 maternal and 773 newborn cases would be prevented, for a net annual reduction of 3365 fewer maternal days and 11 591 fewer newborn days in hospital (Figure 5, Figure 6). If ASA conferred a 10% RRR against preeclampsia and/or intrauterine growth restriction, 151 maternal and 146 newborn cases would be prevented, avoiding 635 maternal and 2187 newborn days in hospital (Figure 5, Figure 6).
Potential impact of a universal acetylsalicylic acid (ASA) strategy on maternal length of stay (LOS) by reducing the number of provider-initiated preterm births (PTBs) as a consequence of preeclampsia (PE) or intrauterine growth restriction (IUGR), based on 2013 data for all of Canada, excluding Quebec. Data are shown assuming that ASA confers a 53% relative risk reduction (RRR) (upper) or 10% RRR (lower) against PE or IUGR. CI = confidence interval.
Potential impact of a Canada-wide acetylsalicylic acid (ASA) program on newborn length of stay (LOS) by reducing the number of provider-initiated preterm births (PTBs) as a consequence of preeclampsia (PE) or intrauterine growth restriction (IUGR), based on 2013 data for all of Canada, excluding Quebec. Data are shown assuming that ASA confers a 53% relative risk reduction (RRR) (upper) or 10% RRR (lower) against PE or IUGR. CI = confidence interval.
Excluding early neonatal deaths (occurring within the first 7 d of life) from our analyses had no effect on any of the above findings (data not shown).
Interpretation
About 25% of singleton preterm births in 2013 in Canada, excluding Quebec, were provider-initiated preterm births. One-third of these were associated with preeclampsia and/or intrauterine growth restriction, and this proportion increased with increasingly preterm birth. Both women and newborns had significantly longer hospital length of stay when maternal preeclampsia and fetal intrauterine growth restriction were present concomitantly, likely a reflection of higher morbidity in these cases. A universal ASA prophylaxis strategy that reduces provider-initiated preterm birth due to preeclampsia and/or intrauterine growth restriction by 53% would avoid 3365 maternal and 11 591 newborn days in hospital, while a 10% reduction would avoid 635 maternal and 2187 newborn days in hospital. The inclusion of Quebec, which accounted for 23% of births in Canada in 2013,28 would further increase the number of hospital days avoided.
ASA, even when started very early in pregnancy, has the same rate of side effects, including gastrointestinal discomfort, allergic reaction, rash and bleeding, as placebo.29 Overall, 0.3%-0.9% of the general population are believed to be intolerant to ASA, manifesting as acute asthma, urticaria and angioedema, for example.23 Certainly, women with a history of ASA intolerance, regardless of their risk of preeclampsia or intrauterine growth restriction, should not receive ASA. But what about women who have no known contraindication to ASA and are at low risk for preeclampsia? We know that recognized risk factors for preeclampsia explain only about 25%-50% of that risk19 and that nulliparous women with no evident risk factors have a 4% absolute risk for preeclampsia.19 The general consensus is that women at low risk derive the same RRR with ASA as do women at higher risk,13 even though the absolute risk of preeclampsia is lower among the former and, thus, the number needed to treat to prevent preeclampsia rises accordingly.19,30 Currently, there is no accurate means to screen for preeclampsia or intrauterine growth restriction.17,18 Certainly, although we do not endorse use of ASA prophylaxis in all pregnant women and reiterate that the strategy should be focused at women at higher risk for preeclampsia and/or intrauterine growth restriction, emerging data about ASA efficacy among women at differing risk may lead to a lowering of the threshold for starting ASA treatment.22
The rate of preeclampsia and/or intrauterine growth restriction that we observed among all provider-initiated preterm births, 33.6%, is consistent with that noted by other investigators.3,4 Morisaki and colleagues31 found that, once preeclampsia or intrauterine growth restriction is present, avoidance of provider-initiated preterm birth is difficult, even when advanced obstetrical care is available. Accordingly, primary prevention of preeclampsia or intrauterine growth restriction is 1 approach to preventing a fairly large proportion of provider-initiated preterm births. Other risk factors for preterm birth, such as younger and older maternal age, twin gestation, and high and low maternal weight,32 are less amenable to intervention. If ASA treatment reduced preeclampsia and/or intrauterine growth restriction by 53%, we estimated that at least 801 cases of provider-initiated preterm birth would be prevented in Canada each year, or 4.6% of the 17 396 preterm births in 2013. This represents an absolute reduction of preterm births from 6.5% to 6.2% among the 269 303 singleton live births and stillbirths in 2013, a substantial reduction in a highly important clinical outcome for which there are otherwise few effective primary prevention strategies.
Meta-analyses of randomized clinical trials suggest that ASA prophylaxis may confer RRRs as high as 89% for preterm preeclampsia and 91% for severe preeclampsia.12,33 We assumed more conservative RRRs for these conditions, in the order of 10% and, at best, 53% but applied these to the general population of women rather than to women identified as being at moderate to high risk for preeclampsia. Although there is emerging evidence that women at higher risk for preeclampsia can be identified,8,10,30 enabling a general clinician to start low-dosage ASA treatment, this strategy does not address all women who are likely to benefit.22
Strengths and limitations
Our results are subject to the assumptions specified in the Methods. However, the degree to which these assumptions affected our results is uncertain. The potential cases prevented by ASA prophylaxis may be overestimated, to the degree that prevented cases of provider-initiated preterm birth do not advance to term birth without preeclampsia or intrauterine growth restriction; provider-initiated preterm birth accompanied by preeclampsia and/or intrauterine growth restriction is not due to 1 of these conditions; and current rates of preeclampsia and intrauterine growth restriction are not actually reflective of the low use of ASA prophylaxis in pregnancy. However, the number of cases prevented was more likely underestimated, owing to our strict definition of intrauterine growth restriction (small-for-gestational-age newborn below the third percentile birth weight), our conservative estimates of the efficacy of ASA and the exclusion of births from Quebec. Although Quebec births were not included, and we did not capture home births (less than 2% of births nationally34), our findings are highly applicable across Canada and account for most provider-initiated preterm births. The data that we used are collated by trained medical record abstractors, and we were able to link maternal and newborn records. As information on ASA intolerance and some risk factors for preeclampsia (e.g., previous preeclampsia) are not captured in the Discharge Abstract Database, we were unable to assess their prevalence within our cohort of women. Knowing the degree to which these factors are present in women who do and do not manifest preeclampsia and/or intrauterine growth restriction could better inform a targeted strategy of ASA prophylaxis.
Conclusion
A universal ASA prophylaxis strategy could lower the rates of preeclampsia and intrauterine growth restriction in Canada and, accordingly, rates of provider-initiated preterm birth. This would likely substantially reduce both maternal and newborn length of stay across Canadian hospitals. However, until there is compelling evidence that administration of ASA to all, or most, pregnant women reduces the risk of preeclampsia and/or intrauterine growth restriction, clinicians should continue to follow current clinical practice guidelines.
Supplemental information
For reviewer comments and the original submission of this manuscript, please see www.cmajopen.ca/content/5/2/E508/suppl/DC1
Footnotes
Competing interests: None declared.
Contributors: Susie Dzakpasu led the study. Susie Dzakpasu, Alison Park and Joel Ray conceived and designed the study. Susie Dzakpasu obtained the data and performed the analyses. All of the authors contributed to interpreting the data and writing the manuscript, approved the final version to be published and agreed to act as guarantors of the work.
Funding: This work was supported by a grant from the Canadian Institutes of Health Research (CIHR). Joel Ray and Prakesh Shah each hold a CIHR Chair in Reproductive and Child Health Services and Policy Research. The funder had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript.
References
- Copyright 2017, Joule Inc. or its licensors