Abstract
Background: The independent French drug bulletin, La revue Prescrire (henceforth Prescrire), annually releases a list of drugs to avoid. The aim of this analysis was to review the status of the 2017 list of drugs in Canada to determine whether they had been approved for marketing, their therapeutic status and whether they have been recommended for listing on public drug plans.
Methods: This descriptive analysis reviewed a list of drugs compiled by Prescrire. The status of each drug in Canada was assessed through the Drug Product Database. Therapeutic ratings were obtained from the Patented Medicine Prices Review Board (PMPRB) and the formulary listing recommendation came from the Common Drug Review (CDR) or the pan-Canadian Oncology Drug Review (pCODR). For drugs without a formulary recommendation the Ontario Drug Benefit (ODB) Formulary was searched to see if the product was listed.
Results: Prescrire recommended not using 92 drugs. The PMPRB evaluated 36 of these drugs; 2 were classed as substantial improvements or breakthroughs, 3 as moderate improvements and 31 as little or no therapeutic improvement. Nine of the remaining drugs that were approved in Canada were not assessed because they were approved before 1988 (the year the PMPRB was established), 4 were approved from December 2015 onward and had not yet been reviewed by the PMPRB, and for 1 the approval date was unknown. Twenty-six of the drugs were evaluated by CDR or pCODR, of which 13 were recommended for formulary listing. Sixteen additional drugs that were not evaluated were on the ODB Formulary.
Interpretation: Many drugs that Prescrire recommended avoiding were available in Canada. The results also highlight the diversity of the conclusions that different expert panels have reached.
Each year the independent French drug bulletin, La revue Prescrire (henceforth Prescrire), releases a list of drugs authorized for marketing in either France or the European Union that should be avoided on the basis of an unfavourable benefit-to-harm ratio.1 Prescrire is a member of the International Society of Drug Bulletins, which aims to promote rational pharmacotherapy.2 It arrives at its decision about the benefit-to-harm ratio by assessing medicines through a multistep evidence-based process. First, it “examines the condition or clinical setting for which the drug is proposed; then the natural course of the disease, the efficacy and safety of existing treatments, and the most relevant outcome measures. This is followed by a systematic search for clinical data on the efficacy and adverse effects of the new drug, and an assessment of the level of evidence. Based on [its] independent analysis of clinical data, [it] form[s] a judgement as to whether or not the new drug is beneficial for patients or whether or not its harmful effects outweigh the benefit.”3 Finally, Prescrire takes into consideration whether other drugs with a more favourable benefit-to-harm ratio are available.
The evaluations produced by Prescrire have been compared with those of drug regulatory authorities in other countries.4,5 This descriptive analysis looks at the status of the 2017 list of drugs in Canada, specifically whether they are available in Canada, their therapeutic evaluation in a Canadian context and whether they have been recommended for listing on federal, provincial and territorial public drug plans. Information about the status of these drugs should be of interest to clinicians in making decisions about whether to prescribe them and to decision-makers in deciding about their formulary status.
Methods
List of drugs to avoid
A complete list of drugs to avoid was taken from the April 2017 issue of Prescrire International, the English-language translation of La revue Prescrire.1 Along with the generic names of the drugs, their benefits and harms were also recorded. The availability of these drugs in Canada was assessed by searching the Drug Product Database.6 If the drug was approved in Canada, the approval date was recorded along with a confirmation that the indication and formulation were the same as those mentioned in the Prescrire article and a notation about whether the drug was available by prescription only or over the counter and whether it was a natural health product. In addition, the anatomic/therapeutic/chemical second-level category for these drugs was determined from the World Health Organization Collaborating Centre for Drug Statistics Methodology (www.whocc.no/atc_ddd_index/).
Therapeutic ratings
Next, for those drugs available in Canada, the website of the Patented Medicine Prices Review Board (PMPRB)7 was searched and its rating of the therapeutic value of each drug was recorded. The PMPRB is a federal agency that determines the maximum introductory price for new patented medications. As part of that determination, it does an independent assessment of the additional therapeutic gain from these medications. PMPRB ratings were available only for drugs evaluated from 1988 (the year when the organization was established) onward. The process used by the PMPRB has been described elsewhere.8 Briefly, the PMPRB’s independent Human Drug Advisory Panel considers 2 primary factors in determining its evaluation: increased efficacy and reduction in incidence or grade of important adverse reactions compared with other medicines. Before 2010, drugs were rated on their clinical value over existing medicines as follows: breakthrough or substantial improvement, or moderate, little or no improvement. Since 2010, the ratings have been slight or no improvement, moderate improvement — primary, moderate improvement — secondary, substantial improvement or breakthrough. In cases where the PMPRB evaluations were not available on the website, PMPRB staff were consulted directly.
Listing on public drug plans
Health technology assessments in Canada are conducted by the Common Drug Review (CDR) and the pan-Canadian Oncology Drug Review (pCODR), both part of the Canadian Agency for Drugs and Technology in Health (CADTH). Canada has no national drug formulary and as a result CDR has made recommendations to federal, provincial and territorial drug plans (except for Quebec) about whether to fund a particular drug since 2003. Since 2011, pCODR has done the same for oncology products. CDR/pCODR accept applications from manufacturers and drug plans and then use expert panels9,10 that consider the clinical evidence, plus input from patients, manufacturers and clinicians, in making their recommendations about whether the plans should list drugs for specific indications. CDR has 4 different types of recommendations: list, list with clinical criteria and/or conditions, do not list at the submitted price (this recommendation was dropped in 2016), and do not list. pCODR issues 3 different types of recommendations: recommend, consider with conditions, and do not recommend. Quebec has its own health technology assessment agency, Institut national d’excellence en santé et en services sociaux (INESSS) (www.inesss.qc.ca/index.php?id=42&L=1), which makes recommendations for listing on the Quebec public drug plan. Decisions by INESSS were compared with those from CDR and pCODR and a κ value was computed.
If there was no recommendation from CDR or pCODR, then the Ontario Drug Benefit Formulary (ODB Formulary) was searched to see if the drug was listed as a benefit, that is, paid publicly, and if so whether the listing was unrestricted or limited use.11 The ODB Formulary lists drugs that are publicly covered for eligible populations in the province of Ontario and was chosen because it serves the largest number of people of any Canadian province, spends the most money12 and therefore is arguably the most influential. Decisions about listing are made by the Committee to Evaluate Drugs following an application by the manufacturer and a submission of the clinical evidence.13 The Committee to Evaluate Drugs has 16 members, of whom 2 are patient representatives. The remaining members are practising physicians, pharmacists and an economist who have expertise in a wide range of specialties including geriatrics, infectious disease, family medicine, pharmacology, health economics, epidemiology and other disciplines.14 The Committee to Evaluate Drugs weighs the clinical value of a drug, its safety profile and its cost effectiveness and considers the recommendation from CDR or pCODR (if available) in making its decision.15 The Committee to Evaluate Drugs then makes a recommendation to the executive officer of the Ontario Public Drug Programs about whether the drug should be considered for public funding and the conditions under which the product should be funded.16
Ethics approval
No patients were involved in this study and all data were publicly available. Therefore, ethics approval was not required by the York University Research Ethics Board.
Results
General characteristics of drugs
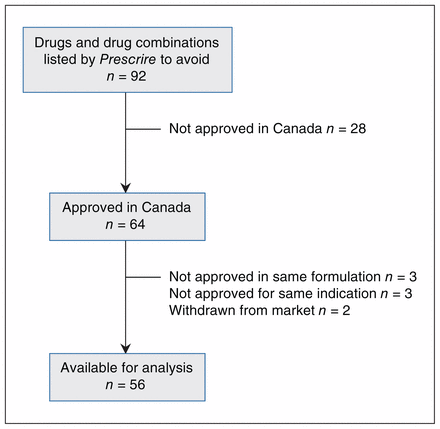
Prescrire listed a total of 92 drugs and drug combinations (88 individual drugs and 4 combination products) of which 64 were approved in Canada (see Supplementary Table 1, Appendix 1, for a complete list of all 92 drugs and all of the data associated with these drugs, available at www.cmajopen.ca/content/6/3/E430/suppl/DC1). However, 2 had been withdrawn from the market, 3 were not approved in the same formulation as described in the Prescrire article and 3 were not approved for the same indication as described in the Prescrire article, leaving 56 drugs for analysis (Figure 1). These drugs were in 24 second-level anatomic/therapeutic/chemical categories, with 3 categories predominating: psychoanaleptics,10 drugs used in diabetes7 and antineoplastic agents.5 Seven drugs were approved before 1980, 3 between Jan. 1, 1980, and Dec. 31, 1989, 9 between Jan. 1, 1990, and Dec. 31, 1999, 18 between Jan. 1, 2000, and Dec. 31, 2009, and the remaining 18 after Jan. 1, 2010; the approval date for 1 drug, glucosamine, a natural health product, was not available. Forty-nine products were available by prescription only, 6 were available over the counter and 1 was a natural health product (glucosamine). Six of the 7 drugs approved before 1980 were over-the-counter medications as was glucosamine (Table 1).
Characteristics of drugs listed as drugs to avoid by Prescrire and approved in Canada
Drug selection.
PMPRB evaluations
The PMPRB evaluated 36 of the 56 drugs. Nine of the remaining 20 were not assessed by the PMPRB because they were approved before 1988, 4 were approved from December 2015 onward and had not yet been reviewed by the PMPRB, and for 1 the approval date was unknown. One drug was not evaluated because it was approved for a second indication (PMPRB evaluates only patented medications for the first approved indication). The reason for the lack of evaluations for the other 5 is not known. Of the 36 drugs that were evaluated, only 2 were classed as breakthrough or substantial improvements and 3 were classed as moderate improvements (Table 2).
Therapeutic evaluations by the Patented Medicine Prices Review Board
CDR and pCODR recommendations and ODB Formulary listing
Twenty-six of the 56 drugs had recommendations from either the CDR or the pCODR. Twelve of these were not recommended for listing and 1 was not recommended at the price submitted by the company. The recommendation for 1 was to list it in a manner similar to other drugs in the class, and 12 had recommendations for conditional listing. There was no recommendation for the remaining 30 drugs, 23 of which were approved before 2003 when the CDR was formed (Table 3). The reason why the remaining 7 were not evaluated is not known. Both CDR/pCODR and INESSS evaluated 21 of the 56 drugs marketed in Canada. The level of agreement in recommendations (κ score) was 0.400, indicating fair agreement (data not shown). Out of the 30 drugs that had no recommendation from CDR/pCODR, 16 were listed on the ODB Formulary (7 were listed for limited use, 8 had an unrestricted listing and 1 was listed as “not a benefit”). Of the 14 that were not listed, 7 were over-the-counter products that are not covered publicly.
Recommendations from the Common Drug Review and pan-Canadian Oncology Drug Review
Drugs with similar therapeutic evaluations from the PMPRB often received disparate listing recommendations from CDR/pCODR (Table 4) and disparate listings on the ODB Formulary (Table 5). One of the 2 products that received a breakthrough/substantial improvement evaluation from the PMPRB was recommended for listing with clinical criteria or conditions and 1 was on the ODB Formulary as a limited-use product. Thirty-one of the 36 drugs evaluated by the PMPRB were rated as providing moderate, little or no therapeutic improvement or slight or no therapeutic improvement but 17 of these were recommended for listing by CDR/pCODR9 or were listed on the ODB Formulary.8
PMPRB therapeutic evaluations and CDR/pCODR listing recommendations
PMPRB therapeutic evaluations and listing status on Ontario Drug Benefit Formulary
Interpretation
The majority (56 out of 92) of the drugs listed as ones to avoid by Prescrire were available in Canada in the same formulation and for the same indication as mentioned by Prescrire. The drugs were predominantly in 3 therapeutic areas — psychoanaleptics, drugs for diabetes and antineoplastic agents — although in total they came from 24 therapeutic areas. Eighteen were approved after 2010. The fact that 7 were approved before 1980, with the oldest one marketed in 1948 (Supplementary Table 1, Appendix 1), reinforces the point made in other studies17–19 that simply because drugs have been used for a considerable period of time does not necessarily guarantee that they are safe. It is also potentially disconcerting that 7 of the drugs did not require a prescription. The 2017 approval date by Health Canada may account for why 2 drugs (defibrotide and vernakalant) were not assessed by either the PMPRB or CDR/pCODR and were not listed on the ODB Formulary.
The results also point to the diversity of conclusions about the benefit-to-harm ratio from different expert panels. These are all drugs that Prescrire said to avoid. The PMPRB seems to be mostly in agreement with Prescrire, at least when it comes to the additional therapeutic value of the 36 drugs it evaluated. Only 5 were assessed as breakthroughs, substantial or improvements or moderate improvements. At the same time, 14 of the 26 products evaluated by CDR/pCODR were recommended for formulary listing, including 9 that the PMPRB said were only of marginal value. The ODB Formulary listed 13 drugs, including 8 that the PMPRB said were only of marginal value.
The difference between the recommendations from Prescrire and the 3 Canadian organizations may at least in part be due to differences in therapeutic cultures. Daemmrich refers to therapeutic cultures as “the historical evolution of a distinctive set of institutionalized relationships among the state, industry, physicians, and disease-based organizations.”20 In addition, the Prescrire caution may be based on differences in the availability of alternative treatments in France and Canada. The differences in the assessments by the 3 Canadian organizations may be due to the fact that they have different mandates: the PMPRB assesses therapeutic value to determine a maximum introductory price, CDR/pCODR assesses therapeutic value in the context of cost-effectiveness and the ODB Formulary also assesses therapeutic value in the context of cost-effectiveness but from the point of view of a payer. Whether these 3 Canadian organizations and Prescrire viewed the same set of studies in reaching their conclusions is not known and this may be a causative factor in the different evaluations. The “fair” agreement between the assessments of the CDR/pCODR and INESSS shows that conclusions of bodies with the same health technology assessment mandate can differ.
Finally, whether the way the 4 organizations deal with conflict of interest leads to differences in their evaluation of individual medicines should be investigated. Prescrire is independent of government and any commercial interests and the experts it consults with cannot have any conflict of interest21 whereas the PMPRB, the CDR/pCODR and the Committee to Evaluate Drugs all manage conflict of interest by requiring committee members to disclose their conflicts and refrain from participating in any decisions where they have a conflict.22–24
Limitations
This study used a secondary analysis of a number of Canadian government and international databases. Those databases have been assembled by reputable sources and have been extensively used by researchers and are regarded as authoritative, but no formal evaluation of their quality/validity has been conducted. Some of the drugs not available in Canada may have been rejected by Health Canada but Health Canada does not release negative decisions. Many of the drugs were not assessed by either the PMPRB or CDR/pCODR and so discrepancies or concordances with the Prescrire avoidance recommendation cannot be assessed. It would also have been extremely useful to have detailed documentation from all of the expert panels to examine why they differed in their conclusions.
Conclusion
There are potentially over 50 drugs that 1 independent expert source recommends avoiding, but in some cases conclusions about these drugs vary. Comparing conclusions may help elucidate why data are interpreted differently. Understanding why these drugs continue to be used is also potentially important in improving physician prescribing and helping to ensure that patients are not adversely affected.
Footnotes
This article has been peer reviewed.
Competing interests: In 2015–2018, Joel Lexchin was a paid consultant on three projects: one looking at indication-based prescribing (United States Agency for Healthcare Research and Quality), a second to develop principles for conservative diagnosis (Gordon and Betty Moore Foundation) and a third deciding what drugs should be provided free of charge by general practitioners (Government of Canada, Ontario Strategy for Patient-Oriented Research Support Unit and the St. Michael’s Hospital Foundation). He also received payments for being on a panel that discussed a pharmacare plan for Canada (Canadian Institute, a for-profit organization) and a panel at the American Diabetes Association and for a talk at the Toronto Reference Library. He is currently a member of research groups that are receiving money from the Canadian Institutes of Health Research and the Australian National Health and Medical Research Council. He is member of the Foundation Board of Health Action International and the Board of Canadian Doctors for Medicare.
Supplemental information: For reviewer comments and the original submission of this manuscript, please see www.cmajopen.ca/content/6/3/E430/suppl/DC1.
References
- Copyright 2018, Joule Inc. or its licensors