Abstract
Background: Oral anticoagulants are commonly used high-risk medications, but little is known about their safety in transition from hospital to home. Our objective was to measure the rates of hemorrhage and thromboembolic events among older adults receiving oral anticoagulant treatment early after hospital discharge compared to later.
Methods: We conducted a retrospective population-based cohort study among Ontario residents aged 66 years or more who started, continued or resumed oral anticoagulant therapy at hospital discharge between September 2010 and March 2015. We calculated the rates of hemorrhage and thromboembolic events requiring hospital admission or an emergency department visit over a 1-year follow-up period, stratified by the first 30 days after discharge and the remainder of the year. We used multivariable regression models, adjusting for covariates, to estimate the effect of sex, prevalent versus incident use, and switching anticoagulants on events.
Results: A total of 123 139 patients (68 408 women [55.6%]; mean age 78.2 yr) were included. About one-quarter (32 563 [26.4%]) had a Charlson Comorbidity Index score of 2 or higher. The rates of hemorrhage and thromboembolic events per 100 person-years were highest during the first 30 days after hospital discharge (25.8, 95% CI 24.8–26.8 and 19.3, 95% CI 18.4–20.2, respectively), falling to 15.7 (95% CI 15.3–16.1) and 6.9 (95% CI 6.6–7.1), respectively, during the subsequent 11 months. Multivariable analysis showed that patients whose anticoagulant was switched in hospital and men had more hemorrhages and thromboembolic events in follow-up.
Interpretation: The first few weeks following hospital discharge represent a very high-risk period for adverse events related to oral anticoagulant treatment among older adults. The results support an intervention trial addressing anticoagulation management in the early postdischarge period.
Oral anticoagulants, including warfarin and the direct-acting anticoagulants, are highly effective for the prevention of stroke and systemic embolism in patients with atrial fibrillation, as well as for the treatment and prevention of venous thromboembolism.1–5 Both conditions increase in prevalence with increasing age.6,7 More than 7 million prescriptions in Canada and more than 37 million prescriptions in the United States are filled annually for oral anticoagulant treatment.8,9 As thromboembolic events increase with increasing age, the absolute risk reduction in events obtained with oral anticoagulant treatment is greater for older adults than for younger people.10–12 Despite their benefit, oral anticoagulants are considered high-risk medications because of the risk of substantial harm — mainly bleeding or thromboembolic events, and death — if treatment is not well managed. 13 Oral anticoagulant treatment has been reported to be the most common drug-related cause of emergency department visits and hospital admission among older adults, with accompanying high mortality rates.14–16
The period immediately after hospital discharge can entail high risk for adverse events, as the transition to home is a complex process involving multiple providers, locations, testing and medication changes with imperfect reconciliation at a time when patients are still recovering. In a 2013 study, roughly one-fifth of Medicare patients discharged from hospital required readmission within 30 days.17 Studies suggest high rates of medication-related adverse events in the early postdischarge period.18,19 Our previous study showed a fourfold greater bleeding risk in older Ontario adults receiving warfarin in the first 30 days after hospital discharge compared to the remainder of the 5-year follow-up.20 Very little is known about the high-risk periods for bleeding or thromboembolic events in the era of direct-acting oral anticoagulant use.
We aimed to measure rates of thromboembolic and bleeding events associated with oral anticoagulant treatment early after hospital discharge (within 30 d) compared to the subsequent 11 months. We hypothesized that the early postdischarge period would be associated with a higher risk of adverse events than the later period. Assuming a medication-focused approach to outcomes as opposed to a disease-specific approach provides a broader view of medication safety.
Methods
Study design and setting
We conducted a retrospective population-based cohort study in Ontario, Canada. All Ontario residents have access to publicly funded physician and hospital care, and those aged 65 years or more also have access to prescription medications with a low or no copay. Study methods and reporting followed Strengthening the Reporting of Observational studies in Epidemiology (STROBE) and Reporting of Studies Conducted Using Observational Routinely Collected Health Data Statement for Pharmacoepidemiology (RECORD-PE) recommendations. 21,22 A detailed protocol with a prespecified analysis plan was prepared and registered at ICES before data were accessed (Appendix 1, available at www.cmajopen.ca/content/9/2/E364/suppl/DC1).
Data sources
Multiple health administrative data sets were linked for this study by means of unique encoded identifiers. Details of the databases and their contents are provided in Table 1.23 In brief, the Ontario Health Insurance Plan database contains billing and diagnostic codes for physician services, the Ontario Drug Benefit program database contains details of outpatient prescription drugs dispensed to those aged 65 or more, and the Canadian Institute for Health Information Discharge Abstract Database and National Ambulatory Care Reporting System detail diagnoses and procedures provided during hospital admissions, and emergency department visits, respectively.
Description of ICES databases used in this study23
We obtained demographic characteristics and vital status from the Ontario Health Insurance Plan Registered Persons Database. We obtained data on cancer, diabetes, congestive heart failure and hypertension from disease-based registries. 24–27 We identified physician specialties from the ICES Physician Database. There is a large literature on the validity and completeness of these population-based databases for identifying drug-related adverse events requiring hospital admission or emergency department visits.28
All diagnoses were coded with the International Classification of Diseases, 9th Revision or the International Statistical Classification of Diseases and Related Health Problems, 10th Revision. We coded procedures using the Canadian Classification of Interventions and the Canadian Classification of Diagnostic, Therapeutic, and Surgical Procedures. Medications were identified through Health Canada Drug Identification Numbers. A list of codes used in the study is available in Appendix 1.
Participants
Eligible patients were those who were 66 years of age or more who started, continued or resumed oral anticoagulant therapy after hospital discharge between September 2010 and March 2015. Anticoagulants included warfarin, dabigatran, rivaroxaban and apixaban. We excluded patients who were in their first year of eligibility for provincial prescription drug coverage (age 65 yr) to avoid incomplete medication records. Patients who had been admitted for a major bleed were excluded, as oral anticoagulant therapy would be contraindicated in many cases. Patients were also excluded if they received more than 1 type of oral anticoagulant at cohort entry or did not have provincial health coverage.
Anticoagulant exposure
There were 4 oral anticoagulants on the Ontario Drug Benefit program formulary at the time of the study: warfarin and 3 direct-acting drugs (dabigatran, rivaroxaban and apixaban). We defined cohort entry as the dispensing date of the first prescription for oral anticoagulant treatment after hospital discharge in the Ontario Drug Benefit program database. This was captured on the day before, the day of or the day after the hospital discharge date. We defined ongoing use of anticoagulant therapy as successive refills of any prescription for an oral anticoagulant within 30 days or 1.5 times the days’ supply of the most recent prescription, whichever was greater. This interval allowed for periodic adjustments to dosages, short pauses and variable timing of refills. If this time frame for refills was exceeded, patients were deemed to have discontinued treatment and were followed for 30 days or 1.5 times the days’ supply of their final prescription, whichever was longer.
We classified eligible patients into incident and prevalent users. Incident users were patients who had not been dispensed an oral anticoagulant in the year before cohort entry, and prevalent users were patients who had been dispensed such a medication in that time. Prevalent users were also classified into 2 groups: switchers (those who were receiving a different oral anticoagulant on discharge than just before hospital admission) and nonswitchers (those who continued with the same anticoagulant after discharge as they had been taking before hospital admission).
Outcomes
Our primary outcomes were hemorrhagic and thromboembolic events requiring admission to hospital or an emergency department visit. We categorized hemorrhages as intracranial, upper or lower gastrointestinal, or other major bleeds based on clinical importance and frequency.29 Thromboembolic events included venous events (deep vein thrombosis and pulmonary embolism) and arterial events (ischemic stroke or transient ischemic attack, peripheral vascular disease embolism and systemic embolism). Multiple studies have established the validity of administrative data for identifying hemorrhage and thromboembolic events.30–36
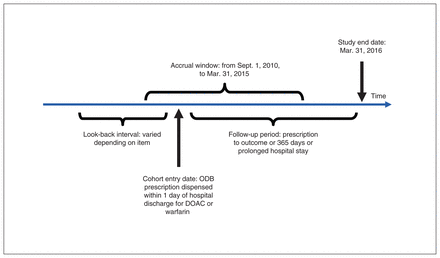
Patients were followed until 1 of the following events occurred: death, discontinuation of oral anticoagulant therapy, hospital stay of more than 5 days for reasons other than hemorrhage or a thromboembolic event, 365 days of follow-up or the end of the study period (Mar. 31, 2016). If a patient had multiple admissions for any outcome of interest during follow-up, we included each event in calculating the rate of events. We assessed major hemorrhagic and thromboembolic events during the postdischarge period at intervals of 0–30 days, 31–364 days and 0–364 days. Figure 1 shows the cohort timeline and definitions.
Cohort timelines and definitions. Note: ODB = Ontario Drug Benefit program, DOAC = direct-acting oral anticoagulant.
Variables
Baseline demographic characteristics included age, sex and rural residence (based on postal code). Data regarding the patients’ care included the oral anticoagulant dispensed at the index prescription date and specialty of the physician who wrote the index prescription. Indications for anticoagulant treatment included atrial fibrillation (emergency department visit or hospital admission for atrial fibrillation within the previous 10 yr); prevention of venous thromboembolism (hip or knee joint replacement within 35 d before cohort entry, or major surgery during the index hospital stay); treatment of venous thromboembolism (diagnosis of acute deep vein thrombosis or pulmonary embolism during the index hospital stay); or active cancer (codes for cancer-related surgery, chemotherapy or radiation in the Ontario Cancer Registry, Discharge Abstract Database or Ontario Health Insurance Plan database within 180 d before cohort entry). Patients could have more than 1 indication.
Other past medical history collected at baseline included hemorrhagic and thromboembolic events within the previous 3 years, hospital admissions in the previous year, recent medications that could interact adversely with oral anticoagulant treatment and comorbidity burden (Charlson Comorbidity Index37). We calculated individual risks of stroke using the CHA2DS2-VASc score, and risks of major bleeding using the HAS-B_ED score (HAS-BLED without international normalized ratio data), employing previously validated database registries.24,38–41
Statistical analysis
We compared baseline characteristics between incident and prevalent users of oral anticoagulants and, within the prevalent users, between switchers and nonswitchers. We calculated crude rates of hemorrhagic and thromboembolic events during the first 30 days after oral anticoagulant therapy was started, 31–364 days after and the entire year. We used modified intention-to-treat principles for the analysis, meaning that events were attributed to the anticoagulant on the index prescription even if the patient switched to another anticoagulant during follow-up. We calculated event rates as the total number of events leading to the hospital admission or emergency department visit for a hemorrhagic or thromboembolic event divided by the person-years available during the interval, stratified by the type of user (incident, switcher or nonswitcher). Multivariable regression models adjusted for sex, age, prevalent versus incident use, warfarin versus direct-acting anticoagulant use, switch versus no switch, indication for use, Charlson Comorbidity Index score, HAS-B_ED score and CHA2DS2-VASc score.
Ethics approval
The use of data in this project was authorized under section 45 of Ontario’s Personal Health Information Protection Act, which does not require review by a research ethics board.
Results
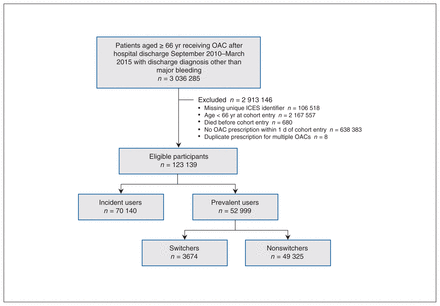
We assessed 3 036 285 patients who were discharged from an Ontario hospital during the accrual period for eligibility. Once exclusions were made for missing identifiers, ineligible age, no prescription for oral anticoagulant treatment within 1 day after hospital admission, death before cohort entry and duplicate prescription for multiple oral anticoagulants, 123 139 eligible patients were identified (Figure 2).
Flow diagram showing participant selection. Note: OAC = oral anticoagulant.
Table 2 shows the baseline characteristics of the cohort and subgroups. The patients’ mean age was 78.2 (standard deviation [SD] 7.7) years. Of the 123 139 patients, 68 408 (55.6%) were women, and 19 931 (16.2%) resided in a rural area. Indications for oral anticoagulant treatment included atrial fibrillation (62 957 patients [51.1%]), recent joint replacement (44 375 [36.0%]), major surgery during the index hospital stay (22 043 [17.9%]), active cancer (7858 [6.4%]), and deep vein thrombosis or pulmonary embolism diagnosed during the index hospital stay (6407 [5.2%]). Patients were most commonly dispensed warfarin (59 232 [48.1%]) or rivaroxaban (51 409 [41.7%]. A total of 70 140 patients (57.0%) were incident users, and 52 999 (43.0%) were prevalent users. Prevalent users were older than incident users (mean age 81.1 [SD 7.6] yr v. 76.1 [SD 7.1] yr), and a higher proportion received their index prescription from their family physician (28 920 [54.6%] v. 12 604 [18.0%]).
Baseline characteristics of older adults in Ontario who started, continued or resumed oral anticoagulant therapy after hospital discharge between September 2010 and March 2015
Of the 52 999 prevalent users, 49 325 (93.1%) were nonswitchers, and 3674 (6.9%) were switchers. A total of 1481 of the switchers (40.3%) switched from a direct-acting anticoagulant to warfarin, and 2193 (59.7%) switched from warfarin to a direct-acting anticoagulant.
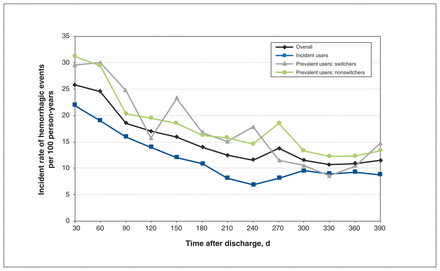
There were 9784 deaths (7.9%) over the year of follow-up. Rates of major hemorrhage per 100 person-years declined from 25.8 (95% CI 24.8–26.8) in the first 30 days after hospital discharge to 15.7 (95% CI 15.3–16.1) over the subsequent 11 months (Table 3, Figure 3). Upper gastrointestinal bleeds were the most common type of specified bleed, with an annual rate of 4.8 per 100 person-years (95% CI 4.6–5.0). Prevalent users experienced a higher overall rate of hemorrhage per 100 person-years than incident users (20.4, 95% CI 19.9–20.9 v. 14.6, 95% CI 14.1–15.1) (Table 4). However, after adjustment for covariates, this difference decreased to an estimated incidence rate ratio (IRR) of 1.01 (95% CI 0.95–1.08). Among prevalent users, the rate of hemorrhage was higher for switchers than for nonswitchers (IRR 1.17, 95% CI 1.02–1.33) (Table 5). Men were more likely than women to experience hemorrhage (21.4 per 100 person-years, 95% CI 20.8–22.0 v. 14.8 per 100 person-years, 95% CI 14.4–15.3), with an IRR of 1.32 (95% CI 1.26–1.40) (Table 6). Choice of anticoagulant (warfarin v. direct-acting) at discharge did not predict the hemorrhage rate (IRR 1.00, 95% CI 0.94–1.06).
Rates of outcome events over time after hospital discharge in the overall cohort
Postdischarge hemorrhage event rates.
Rates of outcome events over time after hospital discharge among incident and prevalent users
Rates of outcome events over time after hospital discharge among nonswitchers and switchers
Rates of outcome events over time after hospital discharge among men and women
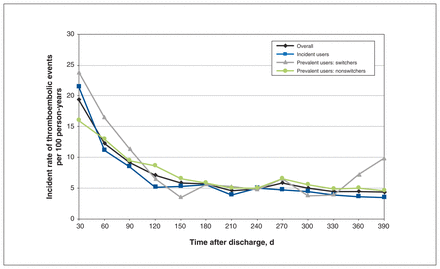
The rate of thromboembolic events per 100 person-years decreased from 19.3 (95% CI 18.4–20.2) in the first 30 days after discharge to 6.9 (95% CI 6.6–7.1) over the subsequent 11 months (Table 3, Figure 4). Of the 4643 events over the year, 2485 (53.5%) were arterial, including 1696 ischemic strokes or transient ischemic attacks or systemic embolisms (36.5%), compared to 1180 deep vein thromboses (25.4%) and 978 pulmonary embolisms (21.1%), representing venous events. The overall rate of thromboembolic events per 100 person-years was higher among incident users than among prevalent users (10.0, 95% CI 9.6–10.4 v. 8.9, 95% CI 8.5–9.2) (Table 4). Although incident users were more likely to have had joint replacement surgery during the index hospital stay and to have lower Charlson Comorbidity Index scores (therefore a higher odds of no events [OR 3.94, 95% CI 3.30–4.69]), those who did have an event were likely to have more events (IRR 3.04, 95% CI 2.75–3.36). After adjustment for the other covariates, switchers were more likely than nonswitchers to have thromboembolic events (IRR 1.30, 95% CI 1.08–1.55) (Table 5). Men had a higher rate of thromboembolic events than women (IRR 1.13, 95% CI 1.04–1.23) (Table 6). Warfarin was associated with more thromboembolic events than the direct-acting anticoagulants (IRR 1.16, 95% CI 1.07–1.26).
Postdischarge thromboembolic event rates.
Interpretation
In this population-based cohort study involving older adults in Ontario, rates of hemorrhage and thromboembolic events were very high in the first 30 days after hospital discharge, considerably higher than in the subsequent 11 months. Although incident users included a large number of short-term users (e.g., venous thromboembolism prophylaxis after orthopedic surgery), their 30-day event rates per 100 person-years were still high, at 21.4 (95% CI 20.2–22.6) for thromboembolic events and 21.9 (95% CI 20.7–23.1) for hemorrhagic events. Prevalent users were more likely than incident users to experience a hemorrhagic event at any time point but were less likely to experience a thromboembolic event. Event rates throughout the follow-up period were significantly higher among men than among women, and among prevalent users who switched anticoagulant agents than among those who did not switch. The mortality rate during the year of follow-up in our cohort, 7.9%, was relatively high.
An overview of event rates from randomized trials in patients with venous thromboembolism showed a rate of recurrent thromboembolism of 1% and a rate of major bleeding of 0.7% at 3 months while patients were receiving oral anticoagulant treatment.42 In patients with atrial fibrillation, the stroke rate without anticoagulation has been reported to be about 4%–5% per 100 person-years, compared to a bleeding rate of 3.5%–3.8% per 100 person-years.43,44 These rates are less than one-third of the early postdischarge rates in our study.
Our results support the contention that transitions in care for patients should be a target for research on interventions intended to lower adverse outcome rates.45,46 A 2014 systematic review showed that frequent patient contact, dedicated teams for discharge planning and home visits were most effective at reducing early readmissions.47 In the Canadian context, a large cohort study showed that rates of 30-day nonelective readmissions and deaths could be reduced with physician follow-up, particularly when the patient is followed by the physician involved in the patient’s hospital care.48 Randomized trials of targeted strategies to reduce readmission in patients discharged receiving oral anticoagulant treatment are still needed and are a high priority.
Sex differences in rates of venous thromboembolic events have been previously reported, although the reasons for higher rates in males are not entirely clear.49–53 Stroke rates in patients with atrial fibrillation may not vary by sex.54 Bleeding rates among men receiving oral anticoagulant treatment, either direct-acting or warfarin, have also been reported to be higher than those in women,53,55 but this has been refuted by other investigators,56 so sex differences in hemorrhage rates in patients receiving oral anticoagulant treatment are also unclear.
Limitations
Our study has several strengths, including a large sample, validated data sources and inclusion of virtually all people aged 66 or more in a large, diverse Canadian province. Limitations include that the results cannot be generalized to younger populations, although transition from hospital care to home is problematic for children as well as adults.57 Second, minor events that do not lead to hospital admission or emergency department visits can still affect quality of life adversely, but are not captured in our data. Third, use of observational data collected as part of routine clinical care always entails some risk of information bias. However, missing data were rare in our study, at less than 0.07%, and misclassification bias for key elements including hospital discharge, prescription dispensing, and morbid outcomes requiring hospital admission or an emergency department visit is known to be low.58–60 We plan to conduct a follow-up study on predictors of our outcome events to address in more detail unmeasured confounding and death as a competing risk of outcomes.61
Conclusion
This study shows that adverse events related to oral anticoagulant treatment after hospital discharge are common among older adults in Ontario, particularly in the first 30 days after discharge. This finding supports the need for trials of organized interventions at discharge and early after discharge, as well as further analyses of predictors of adverse events related to oral anticoagulant treatment.
Footnotes
Competing interests: Anne Holbrook has served as an expert policy advisor for national, provincial and local hospital public drug plans for several decades. No other competing interests were declared.
This article has been peer reviewed.
Contributors: Anne Holbrook conceived the study. Anne Holbrook, Michael Paterson, Diana Martins, Simon Greaves and Tara Gomes designed the study. Anne Holbrook and Harsukh Benipal drafted the manuscript. All of the authors contributed to data acquisition, analysis and interpretation, revised the manuscript for important intellectual content, approved the final version to be published and agreed to be accountable for all aspects of the work.
Data sharing: The data set from this study is held securely in coded form at ICES. Although data-sharing agreements prohibit ICES from making the data set publicly available, access may be granted to those who meet prespecified criteria for confidential access, available at https://www.ices.on.ca/DAS. The underlying analytic code is available from the authors on request, with the understanding that the computer programs may rely on coding templates or macros that are unique to ICES and are therefore inaccessible or may require modification.
Supplemental information: For reviewer comments and the original submission of this manuscript, please see www.cmajopen.ca/content/9/2/E364/suppl/DC1.
Funding: This study was funded by grants to Anne Holbrook from the Hamilton Academic Health Science Centre Alternative Funding Plan Innovation Fund (HAH-16-06) and from the Canadian Institutes for Health Research (365834). It was also supported by the Ontario Drug Policy Research Network, which is funded by grants from the Ontario Ministry of Health (MOH), and by ICES, which is funded by an annual grant from the Ontario MOH.
Disclaimer: Parts of this material are based on data and information compiled and provided by the Ontario Ministry of Health, the Canadian Institute for Health Information, Cancer Care Ontario and IQVIA Canada. The analyses, conclusions, opinions and statements expressed herein are those of the authors and do not reflect those of the funding or data sources; no endorsement is intended or should be inferred.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/
References
- © 2021 Joule Inc. or its licensors